Readers Summary
- What shapes us, genes or environment?
- What is the normal circadian biology in a 24 hour day?
CT-7 is about how we are shaped by our environment by the evolutionary erosion of time that our ancestors faced. All life on this planet is shaped by two major variables in our environment: the sun and the seasonal changes. No matter the place present on earth, there are always alterations in these two factors that are cyclic, and always accounted for by all living organisms in some fashion. In some mammals, like man, it is accounted for centrally in the brain and peripherally in our organ ultradian clocks.
Today, we have proof this is true in the microbiome of man. Our nuclear genes are just not as important as our daily habits in determining our gut microbiome. Mitochondriac wisdom is getting more proof that environment controls our genes. I wonder when researchers will gain this insight? Environmental signals in waveforms affect our mt DNA and our mitochondrial matrix to modulate the function of the nuclear genes. LINK

This is why we have different patterns of aging in certain organs. From an evolutionary perspective, this makes a tremendous amount of sense because life is using the "knowns" of its environment to construct a reality that will ensure its survival. This is the basis of epigenetic signaling that we now know to be the major genetic modifier of the genome of all animals.Organisms do not have life cycles, rather they are life cycles because they are perpetual motion machines harboring subatomic particles that move and change their spin only when life is animated. When the particles stop moving and spinning in them they go from biotic to abiotic and light leaves them and information of organization is lost.
The major signal transducer in Epigenetics is found in the cellular signaling in our cell membranes that interact with the environment and our inner hormones that signal our epigenetic switches sitting on our genes inside the nucleus. Since it is clear that our cold adapted pathways use sensory afferents to signal to open the Ancient Pathway, I think it is time we just have a blog in the CT series that discusses what a normal 24 hour day is like in a human circadian biology.
We will start our day at 6 AM for the sake of the blog. Non Scientists just read only your parts of this blog the first time. We have lots of GEEKS reading this stuff now so I need to hurt their brains so they get the significance of what I am saying from an evolutionary perspective. Neurosurgery geeks have their own separate area because this post has lots of neuroanatomy in it. This should be a review of them and may make them fall asleep—though sleep is a very good thing for those geeks and, more generally, all of us.
How does our day begin and how does it evolve?
Non Scientists: This is the modern warm adapted human circadian cycle:
1. Our brain wakes up with a morning surge of cortisol. That is what turns our brain on at 6 AM. VIP helps do this in long light cycles. VIP is highest at 6 AM and lowest at 6 PM. Ghrelin is also highest in the morning. Ghrelin is an incretin hormone made in the stomach that has a half-life of one hour. NPY and Agouti stimulate the production of ghrelin. Ghrelin sends a signal directly to our pituitary gland and it influences our metabolism. This is why the circadian cycle in the stomach in the morning is critical to optimal health. I laid that out here in this blog and it is an important part of the Leptin Rx reset protocol.
Circadian cycles for the obese are dramatically altered compared to non-obese individuals in the morning. In the normal person, Ghrelin is high when cortisol is highest in the early morning. In them, ghrelin drops fast when food is eaten too. In the obese, ghrelin is much lower in the morning than expected. Moreover, when food is eaten, ghrelin stays elevated for an extended amount of time. This happens because of the inflammation associated with the higher leptin levels in the morning in the obese. Melatonin is known to acutely decrease ghrelin and sometimes in tough cases, I will use supplemental melatonin to demolish the morning ghrelin spikes in people with huge appetites. This is most common in the obese, eating disorders, and in those with a severe leaky gut who crave dairy and carbohydrates. It is also very common for young paleo enthusiasts because of how they embrace blue light technology gadgets of the modern world that destroy melatonin levels in the brain. Ghrelin spikes and stimulates NPY in the hypothalamus increasing our desire and ability to eat a lot more. Leptin makes NPY decline normally, but if one is leptin resistant this does not occur and appetite is out of control at the brain level.
This is why obesity is an inflammatory brain disorder causing hormonal imbalance. Hormone imbalance implies a poor redox potential in different parts of the body. Where the potential is destroyed a certain disease will manifest. Obesity happens when it occurs at the leptin receptor or due to slow energy leak from the inner mitochondrial membrane. This means the obese person is losing energy in black box radiation. It is easy to check but few do with a thermal camera. We see this macroscopically as major alteration is sweating and down-regulation of activity due to an inability to uncouple oxidative phosphorylation at the mitochondrial level. It is not a disease of stress or emotion as medicine is trying to ram down media outlets. It is a problem of an alters the quantum biology of electron/proton tunneling across our proteins.
Moreover, this should explain why the SAD breakfast is so problematic for modern humans. It is marketed as a carbohydrate fest. It is also why the Leptin Rx recommendation for protein and fat are so high in the morning. Protein is the backbone of all life. When we are losing energy and increasing molecular chaos we need to replace our proteins to recapture our balance. It solves this problem fast. I use protein over fat in the Leptin Rx because high-fat levels with low protein in the morning cause a spike in the gastric inhibitory peptide that can induce insulin resistance by itself. I do use high fat in certain cases, like bariatric surgery, eating disorders, hypothalamic amenorrhea, or high EMF levels. Many people do not know this. This is why so many people do not buy Gary Taubes theory of "Why We Get Fat". Gary has only part of the story correct, in my view, because obesity occurs on a spectrum just like autism does because it depends on how the environment affects epigenetic expression. When you understand circadian biology, you get a much more complete picture of how the system works on a 24-hour basis. It turns out electrons control the coupling of biochemistry in life and understanding this helps to make sense of why hormones are disrupted when electrons are not handled correctly. I became a student of circadian biology when I saw the entire view from a 30,000-foot level.
2. At 6:45 AM we will expect to see the sharpest rise in blood pressure in the entire day. This is due to many activated systems in the body getting us ready to fully supply blood to all vital areas to get us motivated to begin our day and search for food. This period of rapid BP rise is why we see so many cardiac deaths occur in early morning sleep or early wakefulness. This happens when cortisol is highest.
3. At daybreak, when the sun hits the retina, the photic stimulus begins to shut off the secretion of melatonin from the pineal gland in the brain. AM sunlight contains mostly IR light at daybreak and as we approach noon, UV light frequencies appear on the skin.
4. At 7:30 AM usually after an hour of light melatonin is completely shut off in the brain.
5. At 8:30 the gut has been awakened and peristalsis becomes more vigorous and bowel movements getting rid of yesterday's food are very likely. This happens by protons flows to move serotonin sulfated by the light of the gut microbiome in the wall to get to the brainstem to become sulfated melatonin. This is stimulated if food is eaten around this time as well. This is called the gastrocolic reflex. Cortisol, aldosterone, and ghrelin are all raised at this time to drive activity, increase our blood pressure and stimulate feeding. This is all yoked to AM sunlight stimulus. It is blocked when we wear clothes or at work in the AM.
6. Around 9-10 AM we have the highest secretions of the sex steroid hormones in humans and these pulsatile crescendos lead to our highest alertness at around 10 AM in our day to allow us to explore our environment.
7. Our ideal muscle coordination occurs at 2:30 PM and this adapts us best to hunt for dinner at this time. An hour later we see our fastest reaction times develop from our motor systems in our CNS.
8. At 5 PM humans exhibit their greatest cardiovascular efficiency allowing for maximal exercising or hunting. This also occurs during a period of time when we have our best rates of protein synthesis in our body. This is why exercise should be optimally done in this window.
9. As the sun falls at 6 PM we begin to see a major change in the cardiovascular system about a half hour later.
10. At 6:30 PM we see our highest blood pressures due to changes in atrial natriuretic factor and antidiuretic hormone (ANF, ADH) in the renin-aldosterone axis.
11. Once this occurs over the next 30 minutes (7 PM) we begin to see a gradual rise in our body temperature as leptin (and IL-6) is released from our fat stores, with agouti's help, slowly after dinner is eaten to signal the brain about our fat mass and inflammatory status.
12. For the next two to three hours leptin levels slowly rise as insulin levels fall. Adiponectin levels also fall during this time frame. These fat hormone signals are what activate adenosine system in our bodies. Adenosine is created over the course of the day; high levels of adenosine lead to sleepiness.
13. This peaks at 10 PM and then the circadian clock allows for melatonin secretion after 3-4 hours of total darkness. Serum leptin is rising quickly now (with agouti's help) as it is released from the fat cells to enter the brain. Agouti is highest at this time of the day, even in a normal person.
14. As these trends continue the GI tract is slowly shut down by the circadian clocks and around 11:30 PM and bowel movements are shut down for the night. This means that the vagus nerve is quiet.
15. At midnight leptin begins to enter the hypothalamus to bind to its receptor in the hypothalamus to signal energy reserves while also yoking energy metabolism to sleep via the hypocretin neurons that control all the sleep cycles. In diurnal animals, sleepiness occurs as the circadian element causes the release of the hormone melatonin and a gradual decrease in core body temperature. This drop in temperature is the stimulus to change sulfated serotonin to sulfated melatonin. This timing is affected by one's chronotype.
16. It is the circadian rhythm that determines the ideal timing of a correctly structured and restorative sleep episode. Melatonin, the hormone from the pineal gland, called the "darkness hormone " is of great importance in the functioning of the SCN. The most important target of melatonin in humans appears to be the SCN, as the SCN contains the highest density for melatonin receptors. A double effect of melatonin in the SCN, namely, an immediate effect and long-term effect, has encouraged its worldwide use against the ill effects of jet lag. This may not be wise to do.
As an immediate effect, melatonin is found to suppress neuronal SCN activity towards night time levels. During the daytime, the SCN neurons must run faster than normal. This is possible because the retina has more DHA in it than the brain. In terms of long-term effect, melatonin can phase shift and amplify circadian rhythmicity of the SCN. Melatonin application has been found to be useful in synchronizing the endogenous circadian rhythms not only in people who suffer from jet lag, but also in blind individuals, patients with dementia, and in shift workers. With seasonal changes in night duration, there are parallel changes in the duration of melatonin secretion, and this leads to more secretion in winter than as compared to summer. In the cold environments of fall and winter, melatonin couples to eNOS and not to light levels. In warm adapted humans in the tropics, the light remains the focus of SCN entrainment year round.
17. After the 4 hours of darkness, melatonin secretion increases and this allows plasma leptin to enter the hypothalamus if we are sensitive to its receptor. If we are leptin resistant, this process can no longer occur.
18. Once leptin enters and binds to its receptors, it affects the lateral hypothalamic tracts to immediately send a second messenger signal to the thyroid to signal it to up-regulate thyroid function and efficiency. This is how we can raise our basal metabolic rate when we are leptin sensitive. These coupled events, matched with leptin's actions peripherally in muscles, occur at the UCP3 sites to burn fat as we sleep at a higher basal metabolic rate.
This means electron chain transport does not make ATP as usual. When leptin allows this uncoupling to occur, we make heat and not energy from normal metabolism. This means we will burn off our excess calories as pure heat. This is one reason why calories in and calories out argument makes no biologic sense once you understand how leptin works. Humans are built to burn fat at night as we sleep to lose excess weight we don't need.
19. The timing of the leptin action is also critical. It usually occurs between 12-3 AM and is tied to when you last ate and how much darkness your retina (SCN) have seen. This generally occurs soon after our hypothalamus releases another hormone called prolactin from our pituitary gland in the brain.
20. The surge of Prolactin is normally quite large in normal darkness but is significantly diminished in artificially lit environments after sunset. This was shown in the Jessa Gamble video HERE.
This has big implications for modern humans. The reason is that prolactin release is coordinated with sleep cycles where autophagy is at its highest efficiency and where Growth Hormone is released. If this is diminished we generally see lower DHEA levels clinically and higher IL-6 levels on cytokine arrays. This is a measure of uncoupling of sleep from normal metabolism. I base every bio hack I do on this step in circadian biology because it is the most important.
21. The normal large circadian prolactin surge we should see at around midnight after leptin enters the brain, does not happen if the patient has leptin resistance, sleep apnea, or has eaten food too close (within 3-4 hours) to bedtime. This blocks leptins ability to enter the brain because of insulin spikes. As mentioned above, this step is usually impaired if you are a post-menopausal female as well. This is often why older women sleep badly and gain weight they can not seem to lose in the gym even with a good paleo template and good habits.
This is another reason I am a big advocate for bioidentical hormone optimization in women. This need is greatest in women who are warm adapted. The need is lowest in the cold-adapted females because their leptin levels are already low due to the cold. Postmenopausal women who are cold-adapted tend to do amazingly well clinically in most disease parameters in my clinical experience. The main problem they face is that their vanity and dogma keep them from using the cold pathways to become rockstars as they age.
Exercise training tends to frustrate postmenopausal women because if their hormone response is altered they have a lot of trouble as they age. Men, on the other hand, do not lose their GH levels until 50-55 years old usually. They are also protected by their testosterone levels which persist throughout life provided that they are not suffering from inflammation which directly lowers their free and total testosterone levels. GH and testosterone keep a mans heart and muscles in tip-top shape. If inflammation destroys these levels earlier in life, it can show up even in younger people. I am finding this clinical result is an epidemic in my own practice.
What happens when step 20 is broken in modern humans?
This commonly happens in diabetics, but it is now becoming a very common finding in modern humans because of the excessive use of technology after sunset. These artificial lights also tend to be quite bright and completely un-yoke the normal circadian signals from the hormone response. Light after sunset reduces the prolactin surge we normally see in humans. When we see chronic lowered prolactin surges we also see lower growth hormone secretion during the anabolic phases of sleep.
Lowered chronic GH secretion directly affects cardiac and skeletal muscle function because the process of autophagy is made less efficient as our life continues. Lowered GH and the sex steroid hormones at sleep lead to loss of cardiac function. This is why heart failure is strongly associated with low IGF-1 and sex steroid hormone levels. When growth hormone is not released in normal amounts, it also decreases our lean muscle mass and increases our fat percentage in all our organs and in our body. This leads to slowly declining organ dysfunction and poor body composition. We can measure this process clinically by looking for falling DHEA and GH/dopamine levels as we age.
What happens in normal aging in step 21?
Aging is among the most common features found in studies on modern humans when DHEA and GH craters on hormone panels. The loss of the prolactin surge is especially prominent in postmenopausal women. Most women begin to suffer from falling DHEA and GH levels around age 35-40 while they are still in peri-menopause. The higher their HS-CRP levels, the faster they enter peri-menopause and the quicker they enter menopause. They also age faster on a cellular level because their circadian chemical clocks are sped up. As a consequence, their telomeres shorten faster as well. Women have higher levels of leptin for childbearing, so they are more prone to leptin resistant issues than men. Leptin is a sexually dimorphic hormone.
This helps explain why older women struggle with cognitive haze, loss of body composition, poor sleep, and increased levels of heart disease after menopause. Many physicians think the losses they suffer are due to the loss of estrogen from ovarian failure, but the loss of growth hormone and progesterone production are far more significant in their physiology. Progesterone is the off switch to anything that is pro-growth. Modern women are usually estrogen dominant even after menopause because of mismatches in circadian biology. Cognitive loss is especially common in post-menopausal women. They also lose on average 1 of their bone mineral density per year from menopause in large part due to the loss of progesterone, not estrogen.
Loss of progesterone also corresponds to poor sleep in these women too. Replacing progesterone in women has a major effect on their sleep and bone stock. It also dramatically improves their memories and cognitive function as well.
Snacking after dinner… Effect on circadian cycles:
If you choose to eat within 4 hours of sleep you will never see the prolactin surge you need, because any spike in insulin turns off this critical sleep time release that corresponds to the cellular maximums of the autophagic process for humans. Agouti, the incretin gut hormone also rises in the blood to higher than normal levels to block leptin from entering the brain.
Diurnal cycles for agouti are coupled to NPY and have major effects on leptin. Agouti is a gene product that normally increases the release of leptin from fat cells at night to signal the brain of what the energy status is of the body. This is great when it is working well. When it is elevated due to heavy carbohydrate use in our diet it creates a massive problem. This is why late night carbohydrate snacking is a bad thing to do.
It appears 12-3 AM are the critical hours at night are where the remnants of mammalian hibernation lie for our species. These are the anabolic times for sleep when we are rebuilding our proteins and recycling our cellular contents. They are three of the most important hours in all human biology. If you miss them, you can bet you have several neolithic diseases for sure. Why do you ask? If these three hours are not reached enough during our sleep cycle, autophagy is never optimized and cellular repair does not occur in our cells. This means we are using old broken down parts in our cells as the next day arrives at 6 AM and cortisol rises again to wake us up.
We can measure this efficiency of this process by checking DHEA and IL-6 levels. I also like to measure hormone panels to see if the inflammation has destroyed any other hormone cascades in aging men or women. This is vital in taking care of older people and treating their longevity. IL-6 levels correspond to Leptin resistant states as well. This makes sleep and metabolic coupling tightly controlled by circadian biology at all times of our life. It is magnified because sleep gets worse as we age and our DHEA, HDL, and HS CRP rise. This is where, during a bio-hack, we can see why circadian mismatches can cause neolithic diseases in humans. Often times we can find the same issues develop much earlier in a young paleo person who has a lot of mismatches in their circadian biology. I test them the same way I would an older person.
Prolactin, Doc?
You must be asking, why is this prolactin hormone so important in a warm adapted human? Prolactin is not just a hormone that secretes human milk. That is the best-known action of prolactin, but not the most important. Immediately after prolactin is released during sleep, another signal is sent to the anterior pituitary to release the largest amount of Growth Hormone as we sleep (GH). GH is stimulated only during autophagic sleep cycles in stage 3 and 4 to increase protein synthesis for muscle growth while you're dissipating heat via the uncoupling proteins. This is where the major release of GH occurs in humans post-puberty when they are warm adapted. 99.9 reading this blog are warm adapted. If you chose to become cold adapted the GH story radically changes, as laid out in CT-6. GH and dopamine are analog proteins.
The implications here are huge for the warm adapted human if this prolactin surge is not adequate to allow us to enter the anabolic stages of sleep. Prolactin surge is diminished by both artificial lights at night and by foods that stimulate NPY, (namely carbs and protein) when they are eaten in fall and winter when biology says they should not be available.

If you are leptin resistant for any reason, have sleep apnea, you will always have an altered body composition because of a low GH level and an altered sex steroid profiles on testing. The reason is that DHEA is the immediate precursor for those hormones and is always low in people with bad sleep efficiency. Most VLCers who are warm adapted face this very problem today. VLC diet is best used in the cold-adapted mammal and not the modern warm adapted lifestyle. In essence, this diet is a mismatch for our modern lifestyle. This is why so many bloggers think ketosis is a dirty word for performance and body composition.
This all implies that as you age you will have higher body fat , lower muscle mass , if autophagy is not optimized by great sleep. This is precisely what we see today in most modern humans as they age. Invariably, their sleep cycles and sleep durations are poor and decreased from their childhood levels. As they age, there is a chronic insidious erosion of circadian biology by decisions made by modern humans over and over again.
What about temperature variations in warm adapted humans?
Where does temperature enter the picture? In warm-blooded animals, homeotherms, such as humans, can change their metabolism in order to keep their heat production equal to the heat loss. Such animals have a temperature control system and thereby maintain a rather constant core temperature. Warm-blooded animals live with the advantage of an unchanged cell activity and temperature in their core. However, the human core temperature falls during the estrogen phase of the menstrual cycle (pro-growth) and during sleep (circadian rhythm by melatonin).
The lowest temperature of the day for modern humans is usually between 2 AM and 6 AM. The temperature cycle is part of the normal circadian periodicity. Our biological clock seems to be synchronized with the rotation of the globe daily. Meal composition and timing, light cycles and temperature play a role in altering normal cycles and autophagic optimization.
Ovulation releases a sharp rise in morning temperature with its estrogen surge. Progesterone effects seem to explain the higher temperature in the last phase of the menstrual cycle where it calms the pro growth effects of estrogen. In post-menopausal women, this balance is usually not ideal, and it leads to many menopausal complaints these women face today.
The reduced temperature induced by melatonin in sleep is needed for Central Nervous System autophagic repair, for another, less well-known reason. The lowered temperature sets the stage for the biologic quantum effects to be optimal on our neurons microtubules that facilitate learning and neuronal spouting that occurs brain-wide.
This is why if you don't sleep well you feel bad the next morning and your mental performance suffers the next few days on cognitive tasks. Research also shows your learning is severely impaired because of lowered BDNF and changes in diurnal cortisol due to the sleep deficit. This is why we monitor truck drivers' and airline pilots' sleep and wake cycles by law!
Moreover, in hospitalized ICU patients or the elderly when this occurs, it sets the stage for the appearance of acute onset delirium. This is exacerbated when they also have a simultaneous cytokine storm from sepsis or obesity. We see this often in hospitalized patients who cannot sleep well in ICUs. Acute delirium states very much look the same as chronic sleep deprivation patients we see clinically as well. Inducing cold, using progesterone and using hypnotics helps manage these conditions. I mentioned this in my hour-long PaleoFX talk last week.
Okay, nonscientists take a breather. Geeks are up: So today we are going to look more closely at how circadian biology sculpts our species. We will assume the sun rises for us today at 6 AM. About two hours before the sunrise we are at our lowest body temperature and this signal is sent to our hypothalamus to the hypocretin neurons that link metabolism (leptin receptor) to the sleep cycle clocks. This temperature dip signals that sleep is coming to an end and that the brain needs to raise its cortisol levels to wake up the cerebral cortex not connected to the autonomic portions of the brain in the brainstem.
This is called the reticular activating system. When the reticular activating system is damaged, humans remain in a sleep-like state called coma. Neurosurgeons call this a chronic vegetative state. The release of cortisol is a neurochemical signal from the hypothalamus that allows the reticular activating system to wake up the cerebral cortex in the AM by increasing water flows from the CSF, Matrix, and cytosol.
Now we have to think about what season we are in? Is a long light cycle (summer) or is a short one that is cold (winter)?
VIP regulates the circadian rhythm in humans and most mammals. VIP is a gut hormone and is found in our taste receptors too! So if we taste the sweetness from carbs in our diet when it's warm and they are growing in the environment, our brain is expecting us to be in a warm season rather than a cold one. So sweet means warm to the brain, not cold. If you mismatch that and eat carbs at the wrong seasonal time, you create inflammation in the brain and it throws off our chemical clocks in our cells and ages us faster. That means our telomeres get shorter. This is not good.
Even geekier: Taste perception and its relationship to glucose homeostasis begin with stimulation of taste cells located in tongue taste buds. There are five basic taste modalities: bitter, sweet, umami, salty, and sour. Taste cells are clustered into taste buds in the tongue epithelium. Mammals have four different types of taste cells (types I, II, III, and IV), exhibiting different molecular phenotypes and functional roles.
Type I cells are glial-like cells that maintain taste bud structure. Type II taste cells transduce sweet, bitter, or umami stimuli and communicate information through G-protein coupled transduction cascades. Type III cells synapse directly with afferent nerve fibers from three cranial nerves and most release serotonin upon depolarization. Type IV basal cells are rapidly dividing progenitor cells that differentiate into type I, II, and III cells. Along with biogenic amine neurotransmitters, it is becoming evident that multiple peptide hormones including glucagon-like peptide-1 (GLP-1), cholecystokinin (CCK), and neuropeptide Y (NPY) as well as VIP are located in taste cells, potentially acting as signaling modulators of multiple gustatory stimuli.
The circadian clock not only can generate its own rhythms but can also be entrained by the environmental light-dark (LD) cycle. Multiple single-cell circadian oscillators that are present in the clock can, when synchronized, generate coordinated circadian outputs which ultimately regulate the overt rhythms.
VIP is a gut polypeptide, has been identified as one of the main neurotransmitters of SCN neurons, and participates in SCN function. These SCN neurons are retino-recipient and are found in the core of the SCN. They are activated by light, and exogenous application of VIP can reset the circadian clock in a manner similar to that of light application, both in vitro and in vivo. It is estimated that 9-24 of SCN neurons express VIP.
Leptin was originally described as an adipocyte-derived cytokine that signals to the hypothalamus to regulate food intake and energy expenditure. Leptin signals through its receptor, which is closely related to the gp130 cytokine receptor. Leptin can induce expression of the neuropeptide gene vasoactive intestinal peptide (VIP) through the VIP cytokine response element, the same element that mediates the response to the gp130 cytokines. Leptin acts synergistically with TGF-beta to activate transcription through this element.
One of the main chemical constituents of SCN neurons is the vasoactive intestinal polypeptide (VIP). Such neurons are retino-recipient and activated by light. Exogenous application of VIP resets the SCN circadian clock in a light-like manner both in vivo and in vitro. These resetting actions appear to be mediated through the VPAC2 receptor (a type of receptor for VIP). Unexpectedly, genetically ablating expression of the VPAC2 receptor renders the circadian clock arrhythmic at the molecular, neurophysiological and behavioral levels. These findings indicate that this intrinsic neuropeptide acting through the VPAC2 receptor participates in both resettings to light and maintenance of ongoing rhythmicity of the SCN.
Neurosurgery geeks only: In mammals, the part of the nervous system responsible for most circadian behavior can be localized to the suprachiasmatic nucleus (SCN). Although previous studies suggest that each SCN neuron may be an independent oscillator, these pacemaker cells must be synchronized to each other as well as to the environment to function adaptively. Therefore, answers to questions about cell-to-cell communication within the SCN lie at the core of understanding how his timing system operates. The daily cycle of light and dark is the dominant environmental cue responsible for synchronizing this biological timing system to the environment. The SCN neurons receive photic information directly from the retinal-hypothalamic tract (RHT). My Vermont 2017 video gets deep into the physics of the retina.
Many of the SCN neurons that receive retinal input from these cells are located in the ventrolateral (or core) region of the SCN and express GABA and, in many cases, vasoactive intestinal peptide (VIP) and the Peptide Histidine Isoleucine. These retino-recipient cells then convey this environmental information to the rest of the SCN. In brain slice preparations, application of VIP alters the firing rate of SCN neurons through a VPAC2 receptor-dependent mechanism and induces expression of mPer1 and mPer2 genes. These two genes are how the circadian cycles yoke directly to the cell cycle and are related to tumor suppressor genes and oncogenesis when mismatches occur chronically in modern man.
Functionally, the administration of VIP, and to a lesser extent PHI, can cause phase shifts of the circadian rhythms in vivo and in vitro in man.
The role of AVP (arginine/vasopressin) in circadian timekeeping has also been well established in the neurosurgery literature. Its role in the control of the circadian rhythm of food and water intake has been reported and well documented. Another intrinsic neuropeptide, VIP, acting through a VPAC2 receptor (a type of receptor for VIP), participates in both resetting to light and maintenance of ongoing rhythmicity of the SCN. NPY and GABA seem to be the neurotransmitters in the projection from the intergeniculate leaflet to the SCN adjacent to CN II. Raphe nuclei projections to the SCN contain serotonin as an NT. AVP and prokineticin 2 are seen in the outputs from the SCN as efferents.
NPY, which is an established neurotransmitter of the geniculohypothalamic tract (GHT), was found to regulate SCN neuronal activity and to produce long-lasting suppression of firing rate of SCN neurons. When co-applied with NPY, NT (neurotensin) was found to dampen the profound inhibitory effect of NPY. So when NPY is high, which would be in equatorial or high light conditions, NPY basically makes the SCN less efficient and allows animals to perform outside their normal circadian boundaries. They stay awake longer for eating and for reproduction in high light times during summer.
All geeks reunite: VIP (along with GRP and AVP) show circadian variations in the level of mRNA in constant contact with environmental conditions from our tongue and our gut. When light becomes long-lasting in summer, NPY dominates the SCN in mammals when light becomes low and the temperature falls to 50-55 degrees constantly at our surface cold receptors, and eNOS rises and blocks all photic input to SCN and circadian rhythms are maintained by a new program. Alpha MSH induces and potentiates that seasonal change within the hypothalamus as laid out in CT-6 blog.
The moral: So the brain is wired for foods when they grow naturally, not when we feel or think we can/should eat them regardless of their availability in modern times.
Leptin sensitivity directly regulates VIP production. VIP regulates the circadian rhythm and entrains the SCN to light. When it is cold, leptin is released from fat cells in large amounts, and we begin to use eNOS to entrain our SCN to cold cycles and we should avoid carbs like the plague then. Remember from CT-6, cold empties fat cells like screaming fire would empty a crowded cinema. In cold, the pituitary-hypothalamic portal is involved in the production of lots of alpha MSH and ACTH. When MSH rises, you are allowing the brain to control everything to get you to optimal. This should make it abundantly clear that cold and warm adapted mammals are not sharing the same circadian biology. Cold selects for supreme LS and superior hormone optimization as laid out in the CT 6 blog.
In long-light summer cycles, when VIP is controlling the SCN again, androgens normalize if the mammal is leptin sensitive. VIP usually fixes our Vitamin D level to optimal too. VIP is a master controller of all inflammation for circadian cycles, but leptin is the hormone that produces VIP in the correct amounts even in light cycles. So if we are leptin resistant for any reason in long-light cycles, we have no control over our circadian cycles and this leads to neolithic diseases.
Normally, VIP lowers our cytokines as the light cycle lessens as the day progresses. At night time the cell is more reduced and not as oxidized. Reduced means better cellular health and oxidized means more cellular inflammation. The act of cellular reduction happens in autophagy during sleep with repair processes. Remember VIP is highest in the morning and this helps it elevate cortisol to wake us up. This is also why cortisol levels are highest when we start our days and lowest in the night when we sleep.
VIP down-regulates most inflammatory cytokines and TGF-B1. It also raises activation of T-regulator cells of our immune system. All of the sickest patients have very low VIP levels and they also have changes in immune toll receptor proteins. Recently, researchers found that circadian rhythms influenced levels of an immune protein called Toll-like receptor 9, or TLR9, causing a daily peak and nadir. I have a sense these are affected by proton spin in cells. When mice were exposed to bacteria at different times of day, those who were infected at the low point of TLR9 activity developed severe sepsis and died much sooner than those exposed when TLR9 was high.
In another phase of the study, mice vaccinated near the daily TLR9 peak had stronger immune responses than those vaccinated at the circadian low point.
This implies those humans with sepsis, leptin resistance, and obesity all have poor circadian cycle control. It should be no mystery any longer why all of these groups suffer from collateral damage from other neolithic diseases. The risk of neolithic disease goes up in all those conditions. This is how mismatches can cause inflammation and shorten our telomeres in our cells. Remember circadian cycles are yoked to the cell cycle as I mentioned earlier in the CT series. This implies that any source of inflammation will shorten our telomeres and speed up our circadian clocks and result in neolithic disease and earlier death.
In the late night (4AM) when temperature levels are lowest, cortisol begins to rise in the blood while leptin levels in the blood are falling. Leptin levels peek in the blood at midnight to 2 AM. They are lowest in the morning when cortisol is rising. When leptin enters the brain and binds, this is the signal that causes the surge of prolactin that allows us to enter the two most important hours of human life when autophagy happens.
Leptin is a direct fatty acid synthase (FAS) inhibitor. Fatty acid synthase is a multi-enzyme protein that catalyzes fatty acid synthesis in humans. Its main function is to catalyze the synthesis of palmitate from acetyl-CoA and malonyl-CoA, in the presence of NADPH, into long-chain saturated fatty acids. Fatty Acid Synthase expression is stimulated by insulin, a hormone produced when blood glucose is high. Fatty Acid Synthase is inhibited by leptin, a hormone that has a role in regulating food intake and fat metabolism. Leptin is produced by fat cells in response to excess fat storage. Leptin regulates body weight by decreasing food intake, increasing energy expenditure, and inhibiting fatty acid synthesis.
Incretin gut hormones geek link: Leptin is an endogenous hormone released from fat cells and it is an appetite suppressant and stops hunger. Hunger is stimulated by the incretin gut hormone called ghrelin that is released from the stomach when light levels are rising and cortisol levels are rising. Recent evidence suggests that food deprivation, and the associated decrease in hypothalamic malonyl-CoA, increases the expression of neuropeptide Y (NPY) and agouti-related protein (AgRP), which stimulates ghrelin release in the stomach to produce the sensation of hunger. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Leptin activates the anorexigenic axis (appetite suppression) in the arcuate nucleus (ARC) of the hypothalamus by increasing the frequency of action potentials in the hypothalamic POMC neurons by depolarization through a nonspecific cation channel and by reduced inhibition by local orexigenic neuropeptide-Y (NPY) neurons.
The cold link: Why CT Simply Rocks
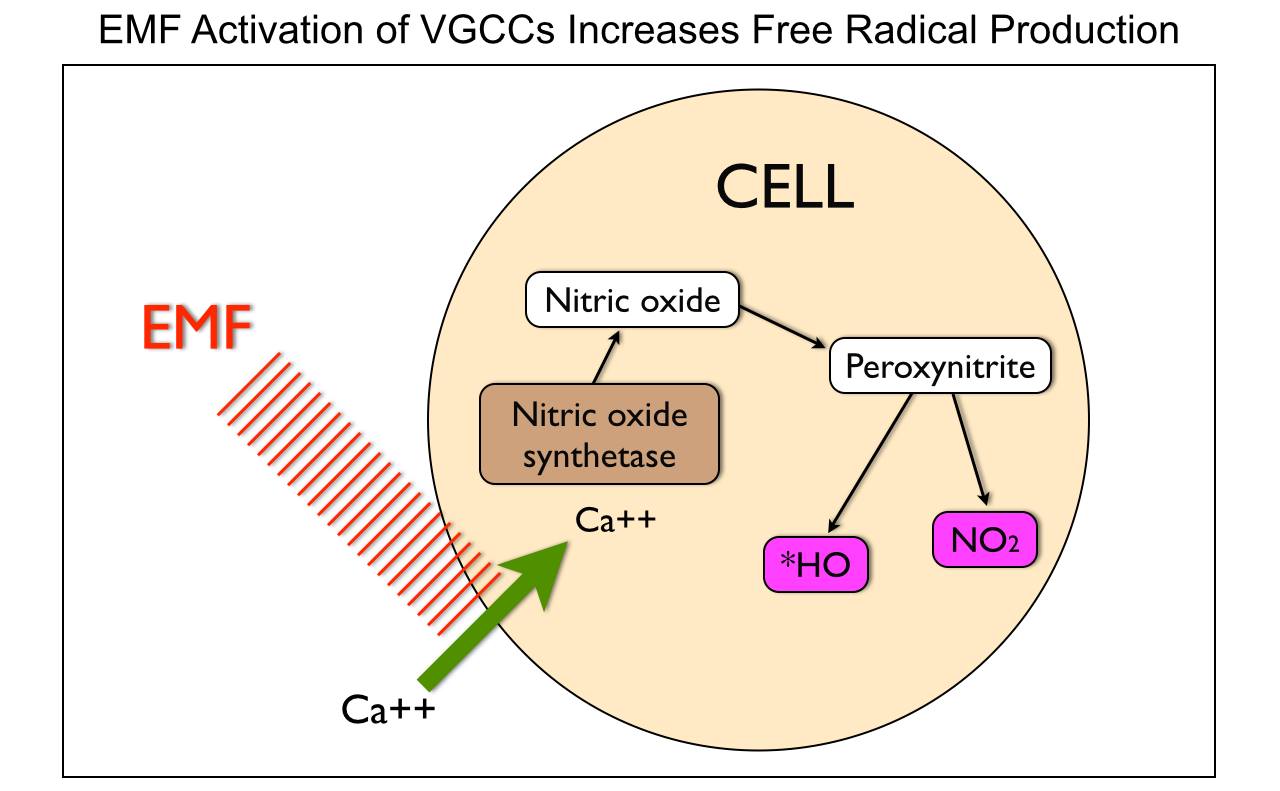
Cold temperatures sensitize us to leptin by causing it to be released from fat cells over time leading to a lower level in the blood chronically. Low temperatures also cause us to increase our RER while eating a low-calorie diet and still maintaining our lean skeletal muscle mass. Hydrogen motions in our matrix are the key to seasonal metabolic rate alternation patterns. These patterns are linked to the power density changes within seasonal EMF's. The native EMF controls the second messenger calcium signals and free radicals inside of cells. Those signals control the flow of hydrogen isotopes in the matrix.
These findings show that during very-low-calorie diets, and low temperatures, a stimulant of a FAS inhibitor, like leptin, and would raise malonyl-CoA levels while decreasing the expression of NPY and AgRP. Clinically, this results in sustained satiation for longer periods of time with less food.
Remember that NPY is also the neuropeptide that is high in the SCN during high light levels when carbohydrates are highest. This peptide is directly regulated by leptin function. So if one is leptin resistant it appears to the SCN that winter has become summer. This is a circadian mismatch and a source of inflammation in the brain. When the cold comes and light drops eNOS is induced and shuts the SCN off to photic entrainment of the circadian clock. The reason for this is not only annual seasonality, but for periodic ice ages mammals have faced on earth and appear to be our primordial situation for life.
This is clearly a survival mechanism that is hardwired into all mammals by evolution, but the ancient pathway has another more important role that we have failed to uncover yet. (FACTOR X)
The environment required for the cold pathway expression is under cold, low light, and low-calorie conditions. All must all be met at once. This is precisely what all cold adapted eutherian mammals are ideally adapted to. These are modern human ancestors, and their biochemistry is foundational to our current paleolithic Ferrari engines. Many believe this pathway represents a starvation response (not), but its real biologic value is of even more interesting. We will talk about this later this year.
This temperature gradient gradually reduces all hunger, pain, thirst, and facilitate sleep in humans and all mammals. These functions were all selected for by evolution via natural selection pressures faced by eutherian mammalian evolution. Moreover, these effects of leptin cause specific epigenetic modification effects on the other hypothalamic hormones or peptides derived from POMC protein cleavage. Those changes are linked via the biology of the POMC neurons in the arcuate nucleus. Leptin and its receptor is especially sensitive to changes in temperature and to the light cycles that humans and all mammals face.
Leptin is intimately tied to hunger, it is linked to thyroid function and directly tied to fat metabolism in all mammals. Fatty acid synthesis, from acetyl-CoA and malonyl-CoA, occurs by a series of reactions that are in mammals catalyzed by individual domains of a very large polypeptide that includes an acyl carrier protein domain. Evolution of the mammalian Fatty Acid Synthase enzyme apparently has involved gene fusion in our evolutionary history. NADPH serves as an electron donor in the two reactions involving substrate reduction. The NADPH is produced mainly by the Pentose Phosphate Pathway. The H in NADPH needs to be a specific quantum spin state.
Every reunites for sleep and immunity: In the warm adapted human
Simultaneously, while sleep is rebuilding our cellular terroir (think levee one), the immune system is also undergoing autophagic repair as well. That is another reason why the temperature has to fall into our bodies. Usually, temperature rises and this causes the immune function to rise and more easily activate in response and duration in fever, stress, and infections. This activation depletes our immune system of its reserves during high light waking hours. Dropping our temperature as we sleep allows us to repair it. During sleep is when the body re-tools our immunity to function optimally the next day. What controls this entire orchestra of hormonal regulation? It's all leptin-mediated and the brain is the master receptive organ to its function.
Sleep is a time for recycling and rebuilding to get us ready for the next day. It is also a time when our immune system is retooled to fight the battle the next day. Wound healing has been shown to be affected by sleep. A study conducted by Gumustekin et al. in 2004 shows sleep deprivation hinders the healing of burns on rats.
It has been shown that sleep deprivation affects the immune system function directly. In a study by Zager et al. in 2007, rats were deprived of sleep for 24 hours. When compared with a control group, the sleep-deprived rats' blood tests indicated a 20 decrease in white blood cell count, a significant change in the immune system. It is now possible to state that "sleep loss impairs immune function and immune challenge alters sleep," and it has been suggested that mammalian species which invest in longer sleep times are investing in the immune system, as species with the longer sleep times have higher white blood cell counts. Rats kept awake indefinitely develop skin lesions, hyperphagia, loss of body mass, hypothermia, and, eventually, fatal sepsis.
It has now been shown that sleep increases telomere lengths on leukocytes in humans. Sleep has also been theorized to effectively combat the accumulation of free radicals in the brain, by increasing the efficiency of endogenous antioxidant mechanisms. These mechanisms are mediated by the hormone DHEA which is the major antioxidant in the brain and correlates directly with effective sleep by lowering IL-6 levels. Progesterone is another critical hormone for brain homeostasis and learning as well. Sleep is vital to mammals, but it is supremely vital to humans because they have shrunk the benefits of hibernation into 2 short critical hours of their sleep cycle because of the massive growth of their brains extinguished the need to sleep through the winter months.
Since man can directly control his environment, therefore, being awake during winter was naturally selected for in his direct ancestors before the primates species because they have the same adaptations. The programs that control our fat mass (leptin) however still remain tied to our ability to sleep well.
Sleep geeks: The homeostatic sleep propensity (the need for sleep as a function of the amount of time elapsed since the last adequate sleep episode) must be balanced against the circadian element for satisfactory sleep. These things are controlled along with corresponding messages from the circadian clock; this tells the body it needs to sleep. Sleep duration is affected by the DEC2 gene. The optimal amount of sleep is not a meaningful concept unless the timing of that sleep is seen in relation to an individual's circadian rhythms. A person's major sleep episode is relatively inefficient and inadequate when it occurs at the "wrong" time of day; one should be asleep at least six hours before the lowest body temperature for optimal functioning. The timing is correct when the following two circadian markers occur after the middle of the sleep episode and before awakening:
- the maximum concentration of the hormone melatonin, and (you can test this but it's hard)
- minimum core body temperature. (Easy to record with new Q self-apps on iPhones)
Sleep Implications:
A University of California, San Diego psychiatry study of more than one million adults found that people who live the longest self-report sleeping for six to seven hours each night. Another study of sleep duration and mortality risk in women showed similar results. Researchers at the University of Warwick and University College London have found that lack of sleep can more than double the risk of death from cardiovascular disease, but that too much sleep can also be associated with a doubling of the risk of death, though not primarily from cardiovascular disease. Professor Francesco Cappuccio said, "Short sleep has been shown to be a risk factor for weight gain, hypertension, and Type 2 diabetes, sometimes leading to mortality.
These all tie to a failure of autophagy in sleep stages 3 and 4 mentioned above. Here, we see why poor sleep links to sleep apnea and the neolithic diseases that are associated with sleep apnea. Growth Hormone is released in a pulsatile fashion from 12-3 AM during restorative sleep cycles 3 & 4, and this hormone facilitates autophagy and recycling of proteins. In essence, GH keeps us younger and in great shape when we sleep like a rock star. The problem is a modern man does not sleep well because of his brain's technology and screen creations.
The metabolic phase during sleep at this time is anabolic which favors repair; anabolic hormones such as growth hormones (as mentioned above) are secreted preferentially during sleep. If things are working well. things get repaired at night as we sleep; and if sleep is poor, repair is either absent or suboptimal. When this occurs chronically, stem cells are used to replace cells instead of using cellular recycling processes that are normally used. Sleep is vital for all our organs rebuilding and retooling.
The human heart fails most commonly by autophagic failure. Heart disease is also the number one killer of both men and women so this means that physicians need to pay better attention to sleep. This is the most important question I assess in an initial bio hack. Autophagy, GH, and sex steroids all strengthen muscle fibers especially that of the cardiac muscle. Here we see the protective role of GH on the cardiac muscle once again.
Using the cold-adapted pathway described in CT 6, is the best way to protect from all circadian erosions, considering we no longer hibernate and have to rely on the two hours of anabolic sleep we get as a replacement. Cold lowers all inflammatory cytokines across the board.
In warm adapted humans, it becomes clear that inflammation is the single most destructive obstacle to human health. This implies that understanding how to control leptin becomes paramount for the warm adapted human.
Temperature variation in menstruating females:
The average temperature falls slightly from infancy to puberty and again from puberty to middle age, but after that stage is passed the temperature begins to rise again, and by about the eightieth year is as high as in infancy. In humans, a diurnal variation in temperature has been observed depending on the periods of rest and activity, lowest at 11 p.m. to 3 a.m. and peaking at 10 a.m. to 6 p.m.
During the follicular phase (which lasts from the first day of menstruation until the day of ovulation), the average basal body temperature in women ranges from 36.45 to 36.7 °C (97.6 to 98.1 °F). Within 24 hours of ovulation, women experience an elevation of 0.15 – 0.45 °C (0.2 – 0.9 °F) due to the increased metabolic rate caused by sharply elevated levels of progesterone. The temperature change is a sign of the movement of hydrogen isoforms moving between tissues in women. The basal body temperature ranges between 36.7 – 37.3°C (98.1 – 99.2°F) throughout the luteal phase and drops down to pre-ovulatory levels within a few days of menstruation. Women can chart this phenomenon to determine whether and when they are ovulating, so as to aid conception or contraception.
Temperature and light have massive biological effects on our biochemistry. We need to be aware of this.
More Support: Webinars by Dr. Kruse
- Factor X (May 2012)
Your Shopping List for this Post
Additional Resources
- Cold Thermogenesis Series
- Cold Thermogenesis 6: The Ancient Pathway
- My Leptin Prescription
- The Quilt: Autophagy
- The Paleo Summit: Is The Paleo Diet The Answer?










First Post? Wow, thanks for continuing with this, quite a lot of work! Trying to get started on this, such a pain to get into cold water.
My morning read with my Bulletproof Coffee (with grassfed butter and whole coconut oil)…. thanks for continuing to education us and to care. I also complete CT Session 4 last night – full body submersion for 30 minutes at 40 degrees. Slept like a baby for 8 hours. First time in many years….. Thank you!
I fall firmly in the non-geek area. My question regarding artificial light in the evening. Is the effect entirely through the eyes, or does light have an impact by falling on skin, hair, etc?
@Obee bright light at the retina ruins the diurnal cortisol circadian cycle from retina to brainstem.
I know Dr. K uses those F lux blue-light inhibitor glasses and/or screen for his computer. Seems largely affected by retina, especially the blue tones.Â
thanks for the cites. it does help to back up when I talk to others since I am an RN.
@Glamazon Cites in this whole series are extensive.
Since there is timing for everything else….is there an optimal time to practice CT?
@Robo not as I have found yet.
I would like to know that, too!
(You’re a rock star Dr. Kruse– thanks for everything) Please clarify: “This temperature gradient gradually reduces all hunger, pain, thirst, and facilitate sleep in humans and all mammals. ” So, as we cold adapt, we naturally reduce our calorie intake? We don’t need to go ‘low-cal’ then start cold adaptation– it will happen automatically?
@Bethany This is absolutely how it works…….this is why calorie restriction occurs normally in cold and is very difficult to sustain in warm…….You are getting it loud and clear now.
Dr. Kruse, Wow. I’m loving this non-geek part. It helps me in understanding more the of the geek parts. I have a couple questions and one not related to this post specifically.
1. You mention the “sweet” taste of carbs telling our bodies it is summer/long light cycles. Would an artificial sweetener do the same thing because of the sweet taste even though there are no carbs?2. I would also like to know if there is an “optimal” time to do CT?
My last question is for a friend’s daughter. I’m not sure if you will have any insight of this, but I thought I’d try.  My friends 4 yr old daughter has just started developing a breast and the doctors think she has Precocious Puberty. They are still running tests to rule out a tumor and to make a diagnosis, but I was wondering if you knew if diet or maybe even some form of CT could help. They said if it is that (and not a tumor) then they want to put her on hormones to stop the early puberty. I think that sounds like a disaster waiting to happen, but I’ve never heard of this before so I don’t know. I would think that messing with hormones at such an early age could cause massive issues as she gets older. Her daughter is lactose intolerant and was put on soy formula as a baby. I don’t know if any of that could be part of the cause, but I’m hoping there is something she can do to help her daughter without the synthetic hormones or surgery. Any suggestions?
@Erica 1. we dont know for sure but I believe artificial sweeteners do affect the entire processes……so I avoid them now.
2. No optimal time for CT …..just do it.
3.Its due to sped up circadian clocks and excessive estrogen……she needs to get tested.
 Hormones are unbound if the liver has to make fat… it has to have grains to make fat… My kid had tons of hormone issues from birth and it turns out she has three elevated risks for liver issues and I think she was unlucky enough to get all three. Although, if you knew about fat being virus storage, I would think she is more evolved than a fat virus carrier. She just has the burden of being near fat. They all three had redundant solutions, no grain. No grain would have solved lots of problems.
 Erica, I swear we are twins separated at birth! I always have the same questions you ask. I was wondering about stevia having that “sweet” effect? Hope not.. Even 99% dark chocolate has some form of sweetener in it–I sure do love my stevia 😉
I know that soy is estrogenic. That is quite messed up though, that’s for sureeeee!!!! So obviously put her on a Dr. K protocol ala kid style. So probably a BAB kid style, omega 3s (in the form of nice fish and seafood). She should also go on the AI (autoimmune) version of LRx kid style too. Clearly she has some kind of gut issues if it’s causing such hormonal issues + the fact she was fed soy. (ruins the gut).Â
That is quite scary to hear about that. Hopefully your friend also will visit this site and read it/implement this protocol. Mother and daughter. Ask Jack about the CT for kids. Usually kids have a bit higher temperature (from infancy to puberty and again decrease from puberty to middle age, then rise again—see in this very post CT 7) and can deal with ‘colder’ temps anyway, but maybe not this girl due to precocious puberty (so might be a bit harder to CT adapt). Make it fun though of course like a lot of swimming in lakes and pools that are naturally ‘colder’, and do winter activities (if this is available where you live) etc.
Dr. K can help best, but that is what I say, b/c I also want to see success for this little girl. 🙂
How does Sex affect the circadian rhythm? If you have it at 9pm or so, will this affect Growth hormone release? Is there an optimal time for? Optimal time not to?
@Kevin Sex in humans is radically different than other mammals…….because of our brain development. That is a future blog. Suffice to say have as much as you can get because it is great for your brain and cytokines.
so, sleeping with my electric blanket all these snowy winters have not been helping me out at all
@jennifer its actually been hurting you.
I am going to buy a stock tank and use that as an affordable large tub. I am 6’2″ and I can not fit in the tub in my house.
http://www.gototanks.com/water-troughs.aspxÂ
@TigerBoy Smart move.
Thank you again for sharing this incredible info with us.
Question: If I am faced with the choice of getting in an hour of CT soak or an hour of sleep – which is better in terms of increasing leptin sensitivity?Â
I feel like if I had a better grasp of all your writing I could answer this myself, but alas, not yet. I am often not home until 10:45pm and although I’m now sleepy earlier since doing the LR reset protocol, I often want to squeeze in a little CT therapy and end up soaking for an hour and not getting to sleep until 11:45pm or midnight if I’m lucky. Then back up at 6am the next morning. This isn’t enough sleep, I’m well aware. But it is my present reality.Â
Am I screwed and there’s no point in doing either, or will CT override the need for sleep somewhat?
@Marie W sleep is always important…….but CT is critical if your sleep stinks. Quality of sleep is more important than quantity of it.
Â
This is my mostest favorite one yet!
I have anosmia from a head injury- how would this affect things regarding the taste section? I also am still overweight and living in a warm climate, but trying to do CT as best as I can using cold baths and ice blocks. Is coconut oil OK? I eat quite a bit of fish and a fairly low carb diet.
@ Anna anosmia will also affect tasting ability……if the neurons are damaged chance of them coming back is small unless you do think the first yr to help……cold, progesterone, DHEA, and a seafood loaded paleo diet.
I’d like to verify the benefits of BHRT (Bioidentical Hormone Replacement Therapy) for postmenopausal women. After menopause I had an annoying short-term memory issue. I would look up a phone number on the computer screen, turn to my phone to dial it and in less than a second the number would be blanked out of my mind. This would happen repeatedly thoughout the day and I attributed it to CRS due to aging.Â
Forward to last year when I saw Dr. K in Nashville for back issues and he referred me to a hormone doc in Atlanta. Within days of starting BHRT my telephone number memory “issue” disappeared. At my 6-month re-check my doc wanted to tweak my dosages and I quickly found out the tweaks were not working when my telephone-number memory issues resurfaced. We revised the tweak and the memory issues disappeared almost immediately. Who knows how many other brain functions have been subtly affected as well?
@Adriana BHRT is critical for post menopausal women and their brains.
Dr. Kruse – will CT and keto-Paleo reverse heart disease? Both my parents have stents and I got my Dad to agree to follow something if there is evidence that plaque build-up can be reversed. I really want to get them healthy but I think it will take some hard science to get him to change. Hopefully it’s out there! Thanks
@Mark in my opinion yes…..but they need to be on Vit D3 and vit K2 and avoid dairy and all grains. Get them to read Wheat Belly to wake them up
Do we scale these times by sunset and sunrise as it changes over the course of the year. For example, since the sun sets at 7:20 here now should we move back #9 and the following steps to corresponding times? I guess this would mean it is optimal to go to bed later during summer hours and get less sleep than the shortest days of winter? Awesome post by the way
@james Clarke yes you can.
Hi Jack, love your work and thank you for making it available for everyone to read. I find it very interesting – have a Bach of App Sc in Nursing. Do you recommend taking Progesterone in peri menopausal women? If so what type brand would you recommend? I would be interested in taking some as my sleep in not the best after 2am. I have reset my Leptin (eat no grains and are on low carb) are 48 years old have a body fat of 11.9% and are not yet “Cold adapted” but thinking about starting. I live in Australia and could swim in the pool at home (gets down to 14/13 degrees in winter), would this help with DHEA and GH/ Progesterone? too?
@Vikkiiquinn It also depends upon case, labs and the context it occurs in……..creams are more common in pre menopausal women but some use oral prometrium. It is a clinical call made after knowing all the variables.
Dr. Kruse, If my understanding is correct from item number 18, it seems that an optimum time to take thyroid meds (Armour) would be at bedtime. Would this be a correct assessment?
@hey Diane It depends upon case…….context is key
Ok, so in the night time, the dipstick for fat stores is the only food one that is really important?
 The protein and carbs disturb the prolactin surge so its stupid to shove them down imo if you don’t want to eat anyway, so where is the fat sensed? in the liver?
The leptin, agouti, and fat … does the fat you split in your liver have leptin?
 The adinosine needs to know about fat where?
I guess I’m just asking specifically where does the fat need to be to sleep, ideally?Â
http://www.bbc.co.uk/news/magazine-16964783 Dr. Kruse did you read this about biphasic sleep being the default? There is a lot of anecdotal support that biphasic sleep works well. I would guess that biphasic sleep with a short phase after dinner and a longer phase around midnight would work well, rather than the biphasic version that has a nap in the early afternoon.
I also have another question, if I may. My gf uses folic acid supplements because apparently when she does not take it she gets very tired and depressed. I doubt she is insulin or leptin resistant, as she’s quite normalweight (maybe a bit on the thin side) and has very limited sugar intake (and ‘normal’ grain intake – at least for the average Dutch person). Trying out any CT is a dead-end (she loathes cold) and I cannot make her eat more paleolike (she dislikes eggs and liver). Reading about folic acid supplements on various paleo sites however does not exactly make me happy about her folic acid ‘addiction’. I wonder if something else is going on. She never had her blood tested, but apparently the folic acid supplements work for her. Maybe some other nutrient is deficient or in too high amounts causing folate depletion? If I could convince her to get a blood test (she thinks it’s not needed – the folic acid works, why bother with it…), what should be a priority to check? (Don’t even think about enumerating 25 tests!) Or if there is a different min/vit to check (magnesium? B complex?) what would it be?
Why does inflammation lower total/free testosterone?Â
Also, I read an article written by Lucas Tafur in which rats centrally infused with beta-hydroxybutyrate (the ketone) into their lateral ventricles showed INCREASED sensitivity both in the brain and in the liver to both leptin AND insulin. In other words, the ketogenic state made them more leptin sensitive. Do you know anything about this, or whether it occurs in humans? The link is here:
http://www.ketotic.org/2011/07/hypothalamic-leptin-and-insulin.html?showComment=1332344886993#c6492203671456509350
@Dan H inflammation blocks leptin from entering brain……it then increases IL-6. IL-6 is chemically also related to leptin……this diminishes DHEA which couple sleep to metabolism and DHEA is the immediate precursor of testosterone. I would also tell you that introducing artificial light to our retina also reduces our insulin sensitivity………without any sugar being involved. That may open some eyes too.
I have read Lucas’s paper……I have a very interesting take……Lucas has not considered yet. When you inject into the lateral ventricle you are on the top side of the thrid ventricle where tertiary order neurons are……the body works the exact opposite way……blood vessels enter from outside in side to the cortex to primary CNS neurons. Lucas and Guyenet always make the same error in thinking the neurological response is the same for Intra Ventricular route and the intravenous route……..they are not. We know this from studies done on HIV patients. That is the topic of a future blog. This is a huge error on assumption………that is just not true.
There is a computer program that is free that changes the spectral range of a computer screen for day and night. I found it here in a comment by Andrea on this blog post about a sleep experiment http://jdmoyer.com/2010/03/04/sleep-experiment-a-month-with-no-artificial-light/#comment-1757
The program is here http://stereopsis.com/flux/
Thanks for that John- I installed it.
Nice…flux. I just installed that on my laptop running Windows 7. It is more mellow on the eyes. Curious to see how it works in daylight now.
@ tiger I have it on everything.
Can you get it for an iPhone?
@Mike Get what?
 Hi Tiger! It is neat. In perfect blue sky daylight here at 1pm aussie time, it is as before, blue balanced to match daylight. The programmer is very accomplished, it seems he did the software for picasa so he knows his stuff. May I encourage you to read JD Moyer’s blog on his sleep experiment too where he eschewed artificial light for a summer month and a winter month, a very gifted and well researched writer.
I have a question concerning the comments. At the top of the blog it currently lists 48 comments but when I look at the bottom I only see 30 listed. Does anyone know the reason that this might be? Also, I don’t see any Dr. Jack comments which is understandable considering how busy the man is but it is unusual. I am just wondering and thank you for any help.
@Larry I see the same thing too and I asked my web folks about it and they said they dont know……I am wondering if it is a MAC issue. I have posted answers to all the questions
something is up…..i have answered every question and I can not see them either. I wonder if it is a MAC issue?
I looked at this page yesterday on my iPhone and saw all your responses there, Dr Kruse, but a minute later they were all gone and I haven’t seen them since
Something has to be up.
Dr. Jack, I wondered that as well but my computer at work is a Dell so I don’t think that is the issue. The odd thing is I can see your response to me but none of your other responses. Thanks again for all that you do. My dad has gone keto in his diet and has begun CT thanks to your website! He has lost 12 lbs & is walking better (spinal stenosis & now amyloid proteins in his one kidney). You are changing the world!
I answered them all on my word press screen but the only way to show up here on discus is to answer here on the blog by signing in with twitter. I may go back to the old format of comments until we figure out why its happening. Ashley we need help its not just Dr. Kruse who cant see me!!!!
 I had similar issues and sent in an email yesterday. I have found that using “Ghostery” to block data tracking by web sites allows me to see all comments, oldest to newest, but in a hard to read format. It also will not allow me to post comments at all. I then discovered that enabling Disqus on Ghostery fixed the formatting issue, but only showed about 2/3 of the actual posts, and annoyingly put them in a random “most popular” order every time by default. It does allow me to post comments, but obviously neither option is ideal at this point. I use Mac and Firefox, if that matters.
@Jack.. Ok good to know. I’m trying to ween myself off of the sweetener, but it’s slow going as I love everything sweet. I’ve gotten rid of everything except for sweetening my hot tea. I’m slightly tea obsessed so that’s a hard habit to change. 🙂
Any specific tests you’re talking about for my friends daughter? She is getting hormones tested to make sure it is Precocious Puberty and of course to make sure it isn’t a tumor next wk I think. I’ve been trying to get her to change her family to paleo and she wants to but gets resistance from her husband and doesn’t have time to research. I try to help, but it’s hard when I don’t know what to tell her for this in simple terms like do this not that. Also, with a kid so young I don’t know how to get her to do CT (they don’t have a pool) and to know how to do it safely with a kid?
When you say sped up circadian rhythms.. does that mean that turning off lights around sunset and blue blocking glass for the girl could help? How do you get rid of excessive estrogen in a 4 yr old? She isn’t overweight she is actually a low weight for her age.
“Jack Says:
March 21st, 2012 at 2:21 pm
@ Anna anosmia will also affect tasting ability……if the neurons are damaged chance of them coming back is small unless you do think the first yr to help……cold, progesterone, DHEA, and a seafood loaded paleo diet.”
Thanks Jack- I know my sense of smell won’t come back due to a left pariatal cranioplasty in the early 1980’s. Just wonder what effect that has since I can still sense sweetness for example but don’t get spice tastes like oregano which are more of a smell. (I once asked an aromatherapy person what effect it might have on me and they had no idea.) I seem to have my sleep apnea licked and am trying to get the most benefit from CT.
Hi from norway, try to follow your advice and eat winter food take cold showers etc, but has now begun to suffer from seasonal allergies now ,especially in the eyes,have you a good advice to this
@Jostein Seasonal allergies tell you that your immune system is not ideally functioning……If you continue CT and maintain good Vitamin D levels and support it with your diet you should notice a big difference as you progress. I used to have this issue and after 3 yrs of this lifestyle change it has completely vanished with out me doing a thing.
Jack, awesome post!
This article came out the same day as this blog, I thought that was cool:
http://www.sciencedaily.com/releases/2012/03/120319163803.htm
Also, I didn’t understand this sentence:
“It is magnified as a human ages because sleep gets worse as we age and our DHEA, HDL, and HS CRP rise.”
Don’t (neolithic lifestyle) DHEA and HDL typically get _lower_ as we age? That sentence reads as though they rise, to me at least.
Oh yeah — I use the Zeo Mobile sleep monitor system.. I’ve been looking back at my food/workout/supplement notes and noticed that taking supplemental Glycine (5g b4 bed) seems to correlate to a higher ratio of deep sleep as well as more total sleep, at least for me and according to Zeo. I quit the Glycine b/c it also seemed to correlate to some stomach upset, but I noticed that there could have been other confounding factors there.
Jack, I’m wondering what your opinions are regarding the importance of Glycine rich foods or supplementation relevant to the subject of this blog?
One other thing, sorry for all the questions but I was wondering also if you could give a summary of the sleep stages you refer to as well. I’m not clear on exactly what those are.
Thanks again for another terrific post..
@Dave if you use the search button on the site and put in sleep I have written about the sleep cycles early on when I began blogging.
Jack,
Any thoughts on the use of a blue light in the morning? I have been using for some time and find it helpful in terms of energy and helping me keep a routine schedule.
http://www.usa.philips.com/c/light-therapy/11625/cat/en/?origin=|mckv|sodvQr4VT&pcrid=8534379174|plid|
@Ethan I have no problems with this.
Dr Kruse,
Does detoxing temporarily increases HS-CRP? Mine went from <0.5 to 5.0? I'm on week 7 of Leptin Rx (Low-Carb Paleo) and week 4 of CT. Had a few detox symptoms? Why else would C-RP go up?
@Ben G there are lots of reasons…….but toxin dump is one for sure.
@ Jostein: With the early arrival of spring in Northern Indiana I have noticed that I am suffering zero from the pollen which usually hammers me pretty hard. I am CTing & doing the LR.
I’m a bit confused about BHRT, menopause and hormones in general. You see, I know several women who used progesterone cream and they gained weight, screwed up their metabolisms etc.
Another question is re: evolution and the use of hormones after menopause. Wouldn’t that not be available, and women living after menopause (b/c it provided fitness for overall family) then how can that be recommended for modern women?
p.s. I just ask these questions b/c I’m interested and am no where NEAR that age to be of concern, but I like to be educated for those around me and for the time when I actually do become that age. Want to be as prepared as possible—like investing long term.
I want to be healthy as possible, for as long as possible.
Thanks.
@Gladina progesterone can make you gain weight if you do not need it……that is why you must be tested and see a doc. There is no room for games.
Thank you for bringing back the old Comments format. Much easier to read. I find the comment numbers very helpful for my note taking. Also much easier to keep track of the new comments. Kudos for acknowledging the issue and taking quick action.
@Atl I felt we had to go back…….Just got finished doing my first CT surgical patient video in the office today.
Dr Kruse,
Here’s the rest of the labs from 3 months ago in December and today…3 months ago until today…I thought some of these numbers would improve quicker, or does the rate of change for 2 month of Leptin reset and 1 month of cold look right? (and the liver ALT(GPT) a detox issue?)
FBG 94 –> 90 (84 at home upon rising on glucometer)
ALKP 95.1 –> 71
ALT (GPT) 36 –> 46 U/L (Detoxing?)
AST (GOT) 21 –> 27 U/L
TC: 193 –> 201
LDL: 126 –> 133
HDL: 33.8 –> 46
Trigs: 168 –> 107
Iron: 178 ng/ml –> 83 UG/DL
B12: 624 pg/ml –>
Folic Acid 1.5 –> >20
VitD 7 ng/ml –> 83.3 (supping 15k-20k IU/day)
TSH: 7.87 –> 4.23
Free-T4: 15.3 pmol/L –>13.40
Free-T3: (didn’t test earlier) now: 4.87
E2: 123 pmol/L –> 104
Testosterone: 219 ng/ml — 336 ng/ml
DHEA-S: 2.71 micrmol/L –> 4.60 (almost doubled)
LH: Now: 2.46 Miu/ML (ref 1.10-9.00)
@BenG HDL rose a lot….that is good. Same thing with DHEA level. Falling TSH also good……..That tells me your liver is doing better and since the HS CRP rose I bet its a toxin clearance issue. B12 is too low……so that maybe why the Crp is up. Increase use of mushrooms, B12, B6 and betaine…….nothing there to me is bad at all. Infact, I think there is a lot there to be pumped about.
Dr. Kruse,
I’ve been taking Solgar B-Complex every day. It has 50 mcg of B-12 a serving. Do I need to go sublingual?
@BenG maybe short term DIM or even IV glutathione would help huge here. Subligual or injectible B12 maybe a lot better.
Also, Iron fell 1/2 even though I’m eating a lot more red meat? Do I need to sup?
@BenG nope.
You mention high fat low protein can induce insulin resistance. What about high fat and high protein (50g+)? Or for Leptin Rx morning meal should you try to moderate fat? I think you had mentioned a 5 egg and 5 pc bacon breakfast was ok before but just wanted to check if there was any concern for quantity of fat in AM as long as protein req is met. Thank you!
@Meghan I have have never seen it once clinically. This is why I use high protein in my reset most often.
Dr. Kruse
Happy to announce yet another weight plateau broken for me. Cutting calories has become effortless – yesterday I was at less than 700 and full all day. Today less than 600 for breakfast (I plan on taking a walk at lunch and skipping dinner again)
I also just received an ice vest and wore it in the car on my way to work to combine with the AC – brrr! still cold and shivering 30 minutes later!
great stuff!
@golooram awesome…….cold selects for calorie restriction naturally
Wow congrats that’s amazing!!
@Everyone READ THIS AND SEND IT TO EVERYONE YOU KNOW. THEY WILL PUT CT ON THE BOOKS IF YOU TELL THEM YOU WANT TO HEAR IT
I need everyone who reads anything I write to go over to the TEDx Nashville site and PM Michael Hart on if you want to hear about the third experiment I completed on January 9th, 2012 that finally got me to launch the Cold Thermogenesis series for you all to learn about and use to regain your health. Go to http://www.facebook.com/TEDxNashville and tell Mr. Hart what you want right now. He is unsure of what to do. Tell him what you want this 3/31/2012. All you Paleo fx folks get in the game today……..
@Jack I think you missed my follow-up question up there.
“Any specific tests you’re talking about for my friends daughter? She is getting hormones tested to make sure it is Precocious Puberty and of course to make sure it isn’t a tumor next wk I think. I’ve been trying to get her to change her family to paleo and she wants to but gets resistance from her husband and doesn’t have time to research. I try to help, but it’s hard when I don’t know what to tell her for this in simple terms like do this not that. Also, with a kid so young I don’t know how to get her to do CT (they don’t have a pool) and to know how to do it safely with a kid?
When you say sped up circadian rhythms.. does that mean that turning off lights around sunset and blue blocking glass for the girl could help? How do you get rid of excessive estrogen in a 4 yr old? She isn’t overweight she is actually a low weight for her age.”
Hi Jack … working on getting you into the TEDxNashville event in 13 days.
Our good friends over at TEDxNashville want to hear from the Dr. Kruse community … in order to fit you into the lineup, they are gonna have to rock the boat. Do you really have something “that important” to share? Why should they make room for you in their lineup? And … what would your community think about you revealing the third experiment at this event (which will give them all a video to share) instead of waiting until later this year?
Let the people’s voices be heard: http://www.facebook.com/tedxnashville
Hey doc, one more question… So my partner and I are on our 3rd week of LRx + CT. She is more Leptin resistant (I’m guessing as she has more belly fat). Now we are at a point where we do 40-50min CT mostly in morning/afternoon, eat dinner around 5 and go to bed at 8-830p (shes normally a bit more tired than I am at this time). I’m starting to wake up early around 4-430a and she has been sleeping so hard she doesn’t wake up til 7a and is still sleepy (and could sleep more). Do you think we need to tweak anything? Do you suppose her body is going thru more change and thus needs the rest? or should make herself wake up early?
Ugh. Still waiting for the whole calorie restriction ct thing to kick in. Got out of the tub and felt like I needed to eat less than a half hour later! Grrr…
@Meghan and Chrystin – are you guys having a high fat high protein meal directly before doing the CT as Dr K suggests? Just wondering…
@krusing Yes we are! Why do u ask?
This is interesting: http://www.ncbi.nlm.nih.gov/pubmed?term=ppar%20agonist%20body%20temperature%20sleep
…7 hours of sleep is way too little for me. Is requiring more sleep (like between 9 and 10 hours) an indicator of some kind of problem? I have vivid dreams, good dream recall, and seemingly excellent health (I’m 25).
Dr. Kruse,
I tried posting this comment before and it seems to have disappeared. Sorry for re-posting. I have been doing CT for over three weeks and followed Dr. Kruse’s protocol exactly. Had no problems with face dunks and spot icing and then took six baths with ice. After my last bath I have the cold uricartia…white bumps on the top of my legs that now are itching after three days. Has this happened to anyone else this far in? Is this the same 03/06 issue that is just happening later? What is the recommendation ? Do I stop until they go away and then start again or do I need to get tested before I start? I hate to stop! I have had no “symptoms” that others have had…it was a very easy transition for me until the hives. Maybe that is not normal either? Any suggestions are so appreciated!
@Julie sounds like a toxin dump of estrogen…..up your B vitamins, consider DIM, or IV glutathione……pretty common once you start emptying fat cells.
@Jack,
please, keep the old format for the comments. It’s simpler and effective -as the old google-, more efficient that the new experiment with a menu and options. The old format saves time, the new format wastes time.
@Santiago – I agree, while it’s nice to see direct replies to comments, it’s hard to filter for new comments and get caught up
Dear Dr. Kruse,
My question is regarding what tests do I really need, and which ones I can skip. Below is a brief history and a pasted list for you to review.
I am an 18 years old very healthy female with no medical issues at all, and have been religiously following the leptin reset diet for 8 weeks now. I have gained 16 lbs while on it. I previously tried the HCG diet, which I followed for 30 days. I lost 20 pounds. However, when I started maintenance, I began to gain inexplicably. Then I found your blog and decided to try the leptin reset.
Most of my life, I have been in the 95 percentile of weight for my age and height. I had my first menstrual period at age 11 (My mom was 13 when she had hers). I started exercising regularly 5 years ago, and lost some weight (amount) slowly but steadily. Then my weight loss seemed to have stalled. So, I stupidly tried a 1500 calorie diet during that time. I was able to lose some weight obviously, but living on 1500 calories or less a day was not easy, which is why I tried the HCG diet. I have 16 pounds I want to lose. I have contemplated trying the HCG again, but with the cold baths to see if this makes a difference. First, however I would like to have the blood tests you recommended to see if there is anything weird going on. As a side note I am doing CT in about a 4 ft long bath. I started it yesterday with 10 lbs of ice (I don’t have a water thermometer yet…) and had no trouble at all, so this morning I did it with 20 lbs. I got in right after dumping the ice into the tube and was warm in less than a minute. About 2 hours after CT on both days, I got a head ache and my stomach began to bother me.
My parents are very encouraging and are willing to go with me to Nashville for a consultation with you if needed. I live in South FL. Please let me know what you recommend.
Below is the list I copied and pasted from your blog. I think I can skip 1,2 and 3. I have already had CBC and it was totally normal. I also had the TSH (1.050 uLU/ml, normal range is 0.400-4.000) and the T-4 Free (1.08 ng/dl, normal range is 0.89-1.62). Do I need to do #5? I don’t drink alcohol. Since I am female, do I need the testosterone tests or the PSA? What is #13, and do I need that based on my age? Also, I am 5ft 7in. tall and right now about 146 lbs though I would like to be at least 134/30.
With gratitude and many thanks,
1. VO2 Max assessment (cardio pulmonary findings lead to a work up if needed.)
2. Initial visit DEXA with body comp. Optimization a DEXA scan to include body composition scores.
F/U visits I use BIA assessments to monitor progress and limit radiation exposure.
3. Neuro cognitive battery of tests to assess processing speed, response speed, memory , learning, and attention repeated annually.
4. Chemistry 20
5. Liver function testing
6. Serum Iron, TIBC, Serum Ferritin and Iron % Saturation
7. Lipid panel: VAP and/or NMR analysis
8. Homocysteine level
9. Complete CBC with differential and platelet count (sometimes an Ivy bleeding time)
10. Hormone panel includes (gender specific)……serum and salivary cortisol, Total testosterone, Free testosterone, % Free T, DHT, LH, FSH, Estradiol (E2) high sensitivity, DHEA-S, PSA, IGF-1, Consider based upon history testing levels of progesterone, pregnenolone, melatonin, serum and urine Osmolarity, Prolactin levels
11. Complete thyroid panel with antibody screening (TPO etc)
12. Complete Urinalysis (women get beta HCG if they menstruate)
13. Highly Sensitive CRP (Not a regular CRP)
14. Complete Vitamin D panel
15. Omega 6/3 serum test (for severe findings consider tissue assay)
16. Fasting serum insulin and HbA1c assessment
@Daydreamer this is the kinda post that make a membership side of the site a must. I cant answer this stuff on the internet. Not kosher for the law.
@ Erica, on searching “prolactin” in pubmed i came across this which might be a starting point for you http://www.ncbi.nlm.nih.gov/pubmed/22404257
Love this post Dr. Kruse! The more I read your stuff, the more I see that living within our biologic constraints and avoiding behavior that causes circadian mismatches is the road to optimal health.
If only the 1st year of med school was living within circadian rhythms-101, paleolithic nutrition-101, optimizing cellular biochemistry-101, with a mandatory ice bath prior to anatomy lab!
Dr. Kruse
I know you spend boatloads of time going through all these posts and I appreciate it.
I know the Kruse hot topic now is CT – and I think it’s great. But if I may I would like to ask an ‘old fashioned’ question about boring old food.
From a nutritional point of view – can you share some thoughts (if any) on raw foods? – specifically proteins and fats.
I find I lose fat faster when I eat this way but I have no freakin’ idea why – would be really curious to hear your thoughts on this as you made a mention of ‘raw lamb leg’ the other day.
Thanks!
@golooram you loose faster because you cant assimilate their macronutients because the food is not cooked. Raw foods are tougher to breakdown and assimilate.
Dr. K, I’m a 56-year-old female that took myself off of BHRT two years ago mainly because the only symptom they helped with were hot flashes. Having been on them for 15 years with many tests and many tweaks and different input from different docs that added up to a lot of money with no insurance coverage I had enough. I found your website in December and started your leptin Rx on January 1. I added the CT when you first mentioned it and have been doing baths about 4-5X/week and icing almost every night. I live in Florida so the weather isn’t exactly conducive to the protocol but I do all the little tricks you mention. And, I’m counting down the days until I leave for my 7500′ elevation with cold rivers in Colorado for three months. Now I am dismayed to hear you say that post menopausal women should be on hormones for optimal. Is this necessarily the case if we’re doing CT? Or, will I need to get on that merry go round of BHRT again? My labs 6 weeks ago showed estradiol and progesterone in the low normal range. My DHEA is low but has been low for years and years – hence lousy sleep (although improving now). And, my testosterone is low and has been for years – so low libido follows. My hsCRP was .4. My four cortisol levels were all in the mid range area. Nothing to brag about here but still trying to wrap my brain around the possibility of BHRT again.
@Sherry it is not mandatory but women who have optimal hormones do fabulously well. The reason you might not have before is because you were not doing the diet or CT then…….when you bring the powers all together this is when the magic occurs.
Thanks so much for answering! Do I continue with the baths or wait for the bumps to go away? I had the red dots all over my stomach at first so an estrogen dump makes sense. I do not feel sick at all…or any of the “symptoms” that others seem to have. I will up my vitamins for sure! Thanks
@Julie up the B vitamins, and or use DIM, and/or glutathione/ NAC to help ……..even feverfew or sillymarin will help.
As usual, fascinating. Jack, is there anyway of making an index (cross linked) from the reader’s summaries. I am spending hours traulling back through when I want to read something again, and can’t quite remember what it was called. Even a simple cross link to each separate blog would be fantastic. I don’t know how you have the energy to keep up with these posts, but do appreciate them!
@Jenny after you hear my opening talk from Paleo fx you will know why…….because I have too.
I agree with Santiago and Mark. The old format (which appears to be back as I write this) may look clunkier, but it works much better.
Sorry to beat a dead horse. But can I start CT again or do I have to wait? Thanks!!!
Dr. Kruse, not to be rude, but you said you had replied to everyone but you didn’t reply to me yet! Looking forward to it.
Dr K…being in the food and restaurant field..the big news in the NYTimes today was that former food critic Frank Bruni has contracted gout..ostensibly from the consumption of too much red meat..at least that is what Bruni claims, going on to cite the most recent anti-red meat study. At some point can you address the red meat debate, its relation to gout et. all…Would be appreciated for those following your ketogenic diet with much success…Thanks
Dr Kruse, I’m a bit confused as to the proper levels of protein vs. fat. My understanding was that too much protein would convert to glucose past a certain threshold. I have been averaging 8% carbs, 28% protein, and 36% fat on a 1450-1900 cal diet. What would be an ideal percentage? PS I usually stay under 30g carbs/day.
@Chris……nope. Protein and fat are perfect marriage. 28% + 36%+ 8%= 72% where is the rest of the 100% there is no other macronutrient class!!!!
Vince, I think there is a lot of evidence out there that fructose causes gout. The red meat studies are junk science. This latest one ia an observational study, which means that at best it might get you an hypothesis, which you could then try to test in a study.
This might help:
http://www.zoeharcombe.com/2012/03/red-meat-mortality-the-usual-bad-science/
@Dr. K,
I completely understand. Thank you for your quick reply.
Sally..the attack on red meat is political..why is there no response from the paleo community
@vince you dont argue with stupid science……you just let it die on the vine. The study was a scientific joke. Those who reviewed this study are being made fun of now.
@Dr. Kruse, Any feedback on question 86? Thank you!
Thanks Dr. Kruse!
I guess I will have to cook more then.
I had dinner before I read this – luckily I had a few roasted marrow bones with my raw Bavette steak.
I hope raw egg yolks are ok though
Vince, I think the paleo community is all engrossed in Dr. Kruse’s ideas. We just don’t have time to get all flustered about yet another silly ‘study’. 😉
@vince the TL/DR is that the paleoshere has shredded so many identical studies like this that it didn’t really register that people would still take these seriously…
That said the community actually did address this pretty comprehensively, MDA even had Denise Minger of “Destroy the China Study” fame do a number on it in a guest post. I think Mark skipped it himself because he was just sick of repeating “Food questionnaires are junk science, and people who use them should be shot” over and over and over and over…..
On that note, here’s Chris Masterjohn on the last (identical one, same authors?) of these studies that was shredded repeatedly: http://blog.cholesterol-and-health.com/2010/09/new-study-shows-that-lying-about-your.html
Dr. Dean Ornish needs to stop putting his approving mark on anything he wants people to take seriously… Seriously…
@Andy I go after Dean Ornish in my book…….he has done more to hurt people in my opinion than help.
Jack, another good read. I have ordered yellow lensed goggles and geenrally find myself sleepy at 9 pm anyway (sun sets at 8 pm now). My sleep is still upa dn down, I generally sleep deeply, but thenw ake between 3-4 am, and stay awake for a while. But I don’t feel anxious or jittery, just relaxed. Then I go back to sleep. I *think* I might be cold adapted. Its cold today, everyone else is freezing, and I’m in t-shirt and shorts, and do not feel cold. Even after dumping 10 lbs of ice around my waist, the skin feels warms within 10 min of removing all the ice (after an hour of CT). I find this all so fascinating… such a strange learning curve!
Dr Kruse,
Is CT more detox and inflammatory causing (> C-RP) than normal weight loss from hypocaloric dieting, because CT causes apoptosis and self-destuctions causes the adipocytes to release all their nasty cargo and inflammatory cytokines which the immune systeme would have to clean up vs. hypocaloric dieting which merely causes adipocyte atrophy and doesn’t release all the nasties?
My other question–on ketogenic CT with regards to the increased strength and V02 max etc. Is the aerobic threshold increased so one can sprint on fat, or does the body increase gluconeogenesis and fill up the muscles glycotic storage more readily to improve anerobic threshold, sprinting? (Or does the ATP-CP pathway improve as well?)
@Andy @Sally…You guys are missing the point …if the NYTimes gives front page space to a misguided food critic and there is no rebuttal then the continued parade of veganism is bound to lead to more disease and obesity…
@drK…Day 12 of CT+Keto….Morning wood completely restored (Age 48)..please include geek and non-geek explanation of this signaling in next post. Men unite.
@Dr. K whoops…typo! Should’ve said 8% carbs, 28% protein, 64% fat. I’ll look to even the ratio of protein/fat. Thanks!
@Chris I have no problem with that ratio at all. Its a great one for winter time. If your hungry on it up the protein…….
Skin temp. In this blog, you say temperature around skin should be 55 degrees, but in the other CT blogs you say we need to get our skin temp down to 55 degrees. Many people are taking this to mean that CT won’t be effective if they can’t measure 55 degrees on their skin while doing CT. I found after a few weeks, it was almost impossible to get the skin under 70 degrees no matter what the exposure due to vascular changes.
So to clarify, the skin just needs to be exposed to 55 degrees (water or air) for CT to take effect?
@Akman best is to get skin temp low……best way to do it is to use submerged water because of conductance. 50 degree water for me is the sweet spot. I wish I lived by the pacific ocean…….its perfect.
Dr. in a previous response to Gladina you said: “@Gladina progesterone can make you gain weight if you do not need it……that is why you must be tested and see a doc. There is no room for games.”
I had my hormones tested on day 19 of my cycle. P/E ratio is not good 61….I was taking 2 DIM daily now down to 1.
Prgesterone 6.5ng (4.4-28)
Estradiol 91.6 (50-150)
Estrone 40.1 (<200)
I lost 6 pounds in the last month doing the LRx, CT & LHT & intervals between 4-5 pm. This was a breakthrough of a 2 year plateau! Started SR T3 a few weeks in and was feeling great(but already was losing weight before the T3) Then I started a small 10 gram dose of Progesterone and have put back on 3 pounds in the last 5 days & am bloated like crazy! I upped it to 20 grams still no relief. I want to be optimal but why would I gain weight when according to my labs I clearly need the progesterone?
@Natural Girl a good doc always pays attn to labs in the context of patients response…….clinical symptoms tell us about what is going on at the receptor level. Id back you down and see how you do for a bit.
Dr. Kruse – quick update on my progress with LRx and CT (in my 3rd week): Improved body comp (weight dropped from 146-137), smashed cravings, improved libido, smashed brain fog, improved skin and hair, and improved mood. Also, I had been having problems with one of my knees after sprinting/basketball before starting your protocols. I coach intramural b-ball, so I played a bit last night (first real exercise since starting), and my knee was completely fine. Amazing! To quote Robb Wolf, “It’s like this stuff really works!”
Quick question: you recommend IV glutathione; what are your thoughts on trans-dermal glutathione? Thanks!
@Evo Mama Im Ok with it but I have to tell you I have had 50% or people love it and 50% of people say it did not work…….So its hit and miss.
@Dr. Kruse – Thanks for following up on Akman’s comment. This morning’s bath was only about ~55F (I guess a couple days of 80F weather will do that to the water supply) and after about 10 minutes it actually felt warm. Shivering was very minor and I felt like I could stay in there forever. I used a bag of ice on my stomach and that was great for getting that area cherry-red, though it only took about 10-15 minutes for it to return to normal after the 45 minute bath, maybe that’s a good sign? Then I started up the alcohol/water ice bag routine again on the ride in to work and that went well. All in all, everything is going great.
@Mark I have to step up CT myself……its gotten really warm. 85-90 already. I am going to buy tons of ice. Looks like this weekend we may launch the membership site on the forum. I need to chillax a bit
Forgot to mention two things:
1) Your comment to Akman helped provide me with some context on what I should be shooting for in regards to water temp.
2) I’m slowing working on submerging my feet more and more, though I’m being careful because the other day I had them in for the entire time (1 hr) and lost some feeling in them and had white spots on my soles and on some toes, which I fixed by putting them in warm water for about 30 minutes. I assume it’s natural for the hands and the feet to take a bit longer to adapt, but I’m hoping that by gradually increasing their exposure that I’ll eventually be able to keep them in the entire time. Word of advice by the way, don’t try to stand up quickly with numb feet. It doesn’t work too well…
Membership site, excellent! What a great birthday present! Now if you get into Tedx Nashville, I’ll have nothing left to ask for! And yes, you definitely deserve some time off.
Vince, there is no rebuttal printed because the NYT doesn’t want one.
@Sally I agree with that
When I went low carb 4 years ago, I was horrified when I developed gout. Oh no, my perfect meat eating diet was not as perfect as all the low carb experts Claim? On the fourth bout it dawned on me that every time I had been away from home and had used processed smoked meats as convent low carb meals. I have found my trigger and I think it has little to do with any good quality meat. Just my 2c.
@Greg I agree…….bad meat is usually filled with grains.
Hi Dr. Kruse,
On the forum you mentioned CT smashing FBG in diabetics; I have certainly seen that in my case.
My question is regarding the OGTT with CT: have you seen this improve in your diabetic patients as well?
I indulged the other night, and had a fairly big spike about one hour out (165!) and then an hour and a half later it was down to 95. Wondering whether it was an anomaly?
Thanks!
Dr. Kruse,
This last post is very helpful. Thanks for staying with us.
I’ve been staying on the Leptin Rx now for 6 weeks. CT using our 55F well water showers and ice packs for other times of the day.
1.)My weight hasn’t budged for a few weeks and I’m getting cold sensations on the top of my thighs. It feels like pins in my leg muscles and is fairly uncomfortable. It only occurs when I first go to bed and put my legs up. If I massage the top of the leg muscles, it seems to diminish and I sleep solidly till sunrise.
2.) At the same time, my bs numbers have been higher than when I first started the Rx. I’m getting 134 2 hours post meal most days and around 114 fasting. What’s going on with my symptoms? I’m stalled at 297 and the diet is solid keto with a lot of fish.
Hope to see you at the TED talks.
BTW, what happened to the Index Tab? Near impossible now to go back and find things you wrote earlier.
Hi Dr Kruse,
In advance, thank you for all your posts. I have found your work very intriguing. Your work puts together an number of streams of research into a big picture.
I have been measuring my morning temperature for over 35 cycles. I have a lowish body temperature. The average is about 36.2C(97.2F) before ovulation and 36.6C(97.8F) after ovulation. Over the last few years I have systematically tried to increase my body temperature to normal. Nothing has worked long term.
I have started measuring my temperature throughout the day and after CT my temperature drop low, like 35.4C(95.7F). Is this a problem? Should I just continue and watch that I don’t go below 35.0C, into hypothermia? Or does this really matter with CT?
Thank you,
Caroline
@caroline dont try to raise it…….its better to be lower.
@vince most of the paleo bloggers that do debunking have already thoroughtly debunked this study, for example….
http://www.gnolls.org/2893/always-be-skeptical-of-nutrition-headlines-or-what-red-meat-consumption-and-mortality-pan-et-al-really-tells-us/
http://healthcorrelator.blogspot.ca/2012/03/2012-red-meat-mortality-study-arch.html
http://www.marksdailyapple.com/will-eating-red-meat-kill-you/
but like you said, it’s political. meaning the mainstream media generally doesn’t touch this side of the argument except to denigrate it.
so what would you suggest be done?
you do understand that we are all wingnuts, right? ; )
Scroll all the way to the bottom below the submit comment box. Index tab appears there.
@Mark I am also trying to immerse my feet more with each soak. My well water is 55 degrees, so that is what I have to work with as I don’t add ice. Yesterday was my 11th soak, so I kept my feet in almost the entire time. I too, wind up with the white feet, but they don’t go numb until I have been out of the tub for about ten minutes. I keep my shoes and socks off for about an hour after so that my feet can warm up. I was reading an article about the “iceman” and I think this was termed “drop off”? Not certain of the term, but I think it might just be part of the process.
Dr. Kruse is this a normal progression with CT, that feet can go numb several minutes after leaving the tub? Thanks.
@Susan normal for me…..i never warm up. I just warm slowly.
@Susan – good feedback, I’m curious to see what Dr. Kruse says. My tap is coming out around 55-60F right now, so we’re in a similar situation it seems.
@Mark — you asked about heart disease reversal and Dr Kruse recommended The Wheat Belly, a book by Dr. William Davis. I might add that Dr Davis has been running a very successful heart disease reversal program for a number of years now, which you can find at http://www.trackyourplaque.com. His approach is consistent with Dr Kruse’s, by and large, but he provides specific detail for heart patients. If you dig into his blog starting with older posts, you will get all the details, or you can get his program guide for to learn the whole picture. I consider myself a successful follower of Dr Davis’s TYP program.
@Gene K – thanks for the follow-up. I haven’t read Wheat Belly but I do know a lot about Dr. Davis. My Dad did read his Track Your Plaque book and he said that there was a part in there that said that if you already have plaque, that there wasn’t much that he could do for you. I haven’t read the passage myself, so I can’t be 100% sure that that is what it said.
@Mark, your Dad’s impression was incorrect. I can share my experience with you offline if you would like to email to me (gene at kalmens dot org).
Dr Kruse, I know you don’t recommend dairy at all. Do you make any exception for aged cheeses, or should they too be avoided? Thank you for your blog. It is transforming my life…
@Gene K for the most part i cut dairy. Only a few raw milk cheeses in Nov Dec form France. That is it.
@Gene K – interesting, I’d love to hear your story. I’ll e-mail you shortly. thank you for reaching out
We had a good lecture here in Cambridge UK about circadian rhythms on Tuesday (2012 Mar 20) by Prof Russell Foster of Oxford University entitled ‘Light, clocks and sleep: the discovery of a new photoreceptor within the eye’ the hour long video is here:
http://www.bna.org.uk/news/view.php?permalink=JT46EDZ325
@Jay people are seeing how important this is. Evolution built this system and we need to live in it.
Gene, with the aged cheeses, my sense is that it should not be a problem depending on the source. I’ve started picking up 1 year aged raw milk Gouda from Holland at Whole Foods on occasion. I would think its a relatively safe and effective source of K2 without the concerns of attaining dairy from American cows. Just a hunch.
@gene Funny you should ask about cheese. Cheese is the one food I could never give up. The weird thing is, my system is violently sensitive to 2 pats of butter but I can eat 1/2# of hard cheese with no ill effects. It is my goto second ingredient of a bag lunch.
@Greg not good for you
@Joe B — I wonder whether due to presence of casein cheeses still cause a leaky gut. I understand they are safe from the carb content point of view.
@GREG Same here, I started eating cheese after many years of not eating them at all, and I love it. Wonder what my next NMR will show…
@GeneK I eat some cheese but very little. Usually I eat them in November around holidays. Herve Mons beaufort cheese…….raw milk french cheese.
@Dr. Kruse – following up on your comment Susan, so it’s OK to have part of the soles of my feet white and some of my toes numb after a tub? Nothing to freak about about, i.e. I won’t lose any toes if I just put some socks on and let them warm up on their own? Once they numbed up, they felt fine, but I started to worry when I saw the white and couldn’t feel some toes, hence why I used the bucket of warm water. How long does it take for the feeling in your feet to come back usually? Thanks, these type of details are greatly appreciated.
@Mark nope. My entire torso has been numb for 8 months now.
@Jack,
for practical, and weakness, reasons I’m not beeing able to go ketogenic as of now. So a couple of carb questions:
1. If one does not get cold adapted, carbs in winter are better than ketogenitc diet?.
2. Is better to be warm adapted and consuming carbs than becoming cold adapted and consume them?.
thx.
@Santiago…..carbs are never good choices in the winter unless you are tropical…….100% It is always better to be cold adapted…….regardless of diet. When the diet is congruent amazing things happen. When the diet is just OK……you win 7 tours or 8 gold medals……..or are great explorer of Everest.
I have to admit Dr. Kruse, water fasting and doing CT is a much steeper adaptation than I anticipated.
@AG it is……its is not for the light hearted. I’m into hour three in the pool now.
@Dr. Kruse – ok, sounds good. Tomorrow morning my feet will be submerged.
Unless you really have been numb for 6 months, (if so, then maybe that’s Factor X lol) is there an amount of time when the feeling in my toes should come back following a tub?
Separately, is it even possible to get frost bite in 50F water? I mean outside of seeing blisters from direct icing (cold utartica), I don’t really have anything to worry about right?
@Mark I have been totally numb for 6 months……..entire torso and part of my limbs. CT inspired. If it is……Frostbite has not happened to me in 6.5 years and I do some pretty deep stuff………but with ice I am careful. I have asked people and most extreme condition doctors told me it is not possible.
That it is not. But pretty impressive results in just a couple days. As a longer term plan, what do you think about doing keto nutrition 5-6 days a week, and water fasting 1-2 days a week?
@AG I do hacks all the time. The biggest one I did was this past January 9th. You will hear about it next week on 3/31 Im unleashing live on the Nashville TEDx stage. This bio hack affected my whole normal plan for this yr.
Water fasting?. Where that has come out?.
@AG is trying something with CT but has not posted anything as yet for results
Am I ALWAYS going to have numb toes after an hour at 50 degree water, or will I eventually adjust? I suffered with plantar fasciitis for twenty years and heels spurs as well. Could this be part of the problem with the toes going numb? Like I said before, they aren’t numb until I have been out of the water for about 10 mins., then they stay that way for quite a while. As Mark needs reassurance, so do I. So, no real problem? Just get used to being cold? Sure hope my ‘furnace’ kicks on sometime soon, so I have and idea if it is working for me or not.
@Susan they should go numb……..the foot issues you have likely will go away with continued CT. I had a bad toe issue that vanished about 5 yrs ago without needing any doc.
I see your response to Mark at comment #172 – Thanks 🙂
I am trying to put myself in a state mind that say that I really ‘enjoy’ sitting in such frigid water. Does mental discipline ever allow for you to enjoy the cold? I do enjoy the fact that I was able to sit in cold water, but when does it become ‘enjoyable’? Ever? Is it going to have to be mind over matter each and every time?
@Susan how I feel after make doing it very enjoyable. the first thing I did in my transformation was master my thoughts……..and I tell everyone it is the most important part.
@Dr. Kruse – Perfect, now I can go forward without fear. Yeah, when I use ice I always spot check periodically to make sure that the skin is cherry red and not white. It’s been fine with ice but not the alcohol combo (probably because that combo gets to about 20F).
Are you going to explain at some point the benefits/reasons for having long-term numbness in your torso? I’m sure the CW response would be that you’ve killed your receptors at the skin but I’m sure that there’s much more to it than that. Thanks again.
@Mark yes……i will explain it as the CT series goes on.
ok, so there has to be an “advantage” of some sort in this numbness state or why would you even go there for a period of 6 months!!!!??? Are you slowing down your internal organs with the cold, like the heart and stomach….AKA
You induced hibernation with CT—for Longevity?
@JIB there are lots of advantages. Longevity is one……but there are others that I can use to for my advantages
@Dr. Kruse – excellent, more to look forward to!
HI Dr. K I have been doing the CT for 34 days on only missed 3 days. The first 3 weeks went well and I was wearing socks in the water. I was seeing improvements in mood and sleep. In the 4th week I added neoprene booties and now I am shivering in the water and not craving the water anymore. Could the booties have something to do with it?
@Kami……stop using modern things to offset the program…..use what mother nature gave you.
Re: circadian cycles…
I just installed the F lux for my computer!! I LOVE it and it’s so easy on my eyes. It really makes a difference and I feel more sleepy but in a nice relaxed normal sort of way. As if it’s a transition, instead of bright light then suddenly going to sleep. Even though I’ve been having good sleeps with CT lately, I think this is just an additional bonus for which I’m grateful for. 🙂
Question: I have been sleeping with a window open for over a month now (regardless of temp. which included a night when it went to -15°C), but I live on a busy street which is loud. I hope that doesn’t ruin things too much b/c sometimes it can be a bit irksome to fall asleep to. I also put towels over my windows (I think since Jan.) b/c there’s too much light from the street light. There’s a slight gap which I figure is OK b/c by the time sunrise comes…I like to see SOME light come through. It’s a tricky combo to get given my current location.
@Gladina f. lux needs to be made for TV’s I dont watch them any longer and I have 13 TV’s in my house. Epic waste of money but I built all this modern convenience when I was busy trying to kill myself as a former obese surgeon……..now I embrace the cold and dark of evolution…….and my biology has responded gorgeously.
Just do what you can to get the cold and dark………your body will do the rest of the magic.
You have been entirely numb in torso and limbs for 6 months?? Is it uncomfortable? I have to admit this really scares me. Are you ever going to get your feeling back?
@Brenda…….nope……..very enlightening actually. My feeling does come back when I allow it…….but I dont want it too just yet. I am hoping by August I will be ready to let it come back.
Dr K, even though I have not yet started your recommended CT regimen, I started taking cold showers – in the morning, after gym, and before going to bed. Is there a benefit in this minimalistic approach or will it really not make any difference? While I can now stand cold showers pretty easily, I started waking up 3 hours into my night sleep, with the rest of the night ruined. I changed my cold only bedtime showers to cold first and warm next to see these unwanted wake-ups disappear. Is it an expected consequence or is there something else going on? Thank you!
@Gene K nay amount of CT is good…….the more we do the better because it is how we fine tune our Ferrari engines that mother nature gave us.
I just don’t get the point of being numb.
@Pam. You will soon enough.
Dr. K .. Could I declare myself a starfish or only you has that authority? 😀
I have a question regarding sleep and cold. You never mentioned anything regarding how to sleep in the CT series or elsewhere. Yet, I see discussions regarding sleeping with window open and such. Cold hibernation time.
I can not figure this out. I can sleep in a very cold room but I need to have heavy covers. Light covers prevent the deep sleep. Not well rested feeling is what I get if covers are light or if I get cold when asleep.
Could you please elaborate on this so we know where to sleep? Under or over the covers? 🙂
Cooling
@Cooling if you want to sleep like a rockstar make it cold and dark………on top of the covers naked. it takes practice but I do this all the time……..it took me about 6 months to get to that point in 2007.
Dr. K .. You remarked once that exercising while doing CT is more beneficial than CT in a tub.
Since I can no longer get cold water from my faucet into the little tub I have I started swimming in 17c = 62f water (Mediterranean). been doing it for 2 weeks now
I am doing about 30-45m 3-5 times a week weather permitting.
I am in ketosis. 6.2, 205lbs.
1- My goal is to regain IR and get FBG into 80s (100s now)
Could you please explain the benefit of such a setup?
2- Any estimate time wise till IR reversal? (weeks months years)
3- You CT for hours in your pool but this is open water swimming. Optimally, How long to stay in water?
Gratefully yours,
Cooling
@Cooling 1. That should happen in a few months assuming your liver is OK……and your eating no oxidized PUFA or tons of carbs…….and your liver is LS.
2. With out detailed testing no way to estimate but it wont take long with the diet and CT………remember what I did in 11 months……133 lbs of change.
3. now I am doing 2-5 hours……because I am trying to reverse somethings I did in a bio hack I did in Jan.
Dr.K, I took a bath around 53 degrees for 40 minutes in the evening and got really sick last night, 5 hours of puking and running. I have a fever too. Could this be a part of detox or just a coincidence?
@Mart probably coincidence…..or you must be loaded with toxins! IF your HS CRP was 10 i’d blame it on toxins………lol
Can’t wait for tomorrow…will be having oysters w/ the bf!
Hmmm…you say ‘trying to REVERSE’ something you were bio-hacking in Jan. interesting…Like maybe you did something similar to what Wim Hof did with the bacteria injected into him with him not getting sick.
I am looking forward to TEDx Nashville to know exactly what you did!
Thanks Dr. K.
Dear Dr. Kruse,
I have been having very beneficial results with your CT/VLC therapy, and I have been trying to convert the family. My wife is coming around, but my 2 sons (9 & 11) are in the “NO WAY DAD” zone.
Sometimes I think my family thinks I am going nuts, by doing all these things to my body.
Your wife and family must be VERY supportive/understanding. She knows of the suffering you had when you were overweight. And now she must see the fulfillment you have from uncovering mother natures “secret”.
BTW does FACTOR X have to do with Vit K and thrombosis?
The Best
@Conan try not to convince anyone……when the student in them appear the teacher in you then should appear and not a day sooner. Factor X has nothing to do with this……..
Just got done with CT with no booties and I did not shiver while in the water and I recovered more quickly after I got out of the water. Now to train my brain to not get in the way of healing.
@Kami…….you learn fast.
So my partner and I are on our 3rd week of LRx + CT. She is more Leptin resistant (I’m guessing as she has more belly fat). Now we are at a point where we do 40-50min CT mostly in morning/afternoon, eat dinner around 5 and go to bed at 8-830p (shes normally a bit more tired than I am at this time). I’m starting to wake up early around 4-430a and she has been sleeping so hard she doesn’t wake up til 7a and is still sleepy (and could sleep more). Do you think we need to tweak anything? Do you suppose her body is going thru more change and thus needs the rest? or should make herself wake up early?
@Meghan just keep it up…….all sounds good.
Dr K – previously you mentioned that a patient of yours loved a cooling blanket. I have found both cooling blankets and cooling pads for beds. Any preference as to which item would be better for helping stay cool while sleeping?
@ATL it is dealers choice…..everyone has their own proclivities they enjoy.
http://www.youtube.com/watch?v=1N_9HZRVl-g&feature=share y’all take a look see here…….next week I am elevating the CT game for what humans can do.
“F.lux for TV” got me thinking and I just finished adjusting the color temp, RBG, saturation, backlight, etc. settings on the TV & voila: no more bright blue screen. Whether or not it lets me sleep easier remains to be seen.
@Kathy I gave up on TV a few years ago……..I have googles but I am so into the dark now I wont go back…….
Hi Dr. K–I have a question about eating. I feel fine before and after lunch, but come 4:00pm I start to feel tired and nauseous. I hold off and don’t eat until 5:00. Then I feel better after eating. Is this LR at the liver level? Or low stomach acid? Or something else.
@Kami my bet is that your body is re adjusting to the new signals you are giving it………it should moderate after a few weeks……we adapt awfully fast. Much faster than we ever knew
CT question — I have notice some interesting events happening the last 35 days of doing CT. An old sun spot appeared on my nose and then disappeared in about 3 days. I used to get another spot on my nose that would bleed (about the size of a pencil tip). That re-appeared without bleeding and then disappeared within 3 days. Now my ears are hurting like they did 8 years ago. Am I healing backwards? Did you see this in your journey?
@Kami I did……I had a lot of skin manifestations because of the sun damage and estrogen dumping……but all passed. I did use a lot of methylators back then when I was doing it……People with FM and lyme have to be real careful ……..because their fat is loaded with toxins.
I like to see the healing. Saw the Doc in CA and he said looks like Lyme is not a player for me. That is good news. Based on Igenex test and the way I responded to the antiobiotics I took. We shall see. I also do have FM and I have high mercury but the 2 1/2 year treatment I had done was too mild. He happened to know the Doc I saw in MN for this and he has been through my town in SD. Small world. He is starting with a Phosphatidyl Choline push. I need a doctor here to partner with me and the CA Doc. Looks like I might have found one. I will find out Mon or Tues. He said he is totally CW but would consider partnering since he would have directions from the CA Doc. Also did a lot of viral testing. Looks like I have 5. Not sure what he will do about that. Maybe just start the Shoemaker protocol. I am waiting for 2 more tests. Methylation and the nose staph infection test (I can’t recall the name). I consult with them April 5 to see the plan. CSM for now which is going fine. Just once a day. I am anxious to start as I feel my body will heal fast while doing the CT. I gave him CT 6 to read.
@Kami you should look in to the PKT protocol of Patricia Kane PHd. See if anyone out your way can do it. I have a person here in Nashville that does it.
Would taking DIM help me with detox at this time? I do supplement estrogen because of being low and my body does not like progesterone at this time.
@Kami it would if estrogen is the issue.
Not sure if estrogen is an issue, but I always carry fat on my thighs that doesn’t seem to budge. It looks so out of place on my body. My nemises. I am hoping CT will change this. My Doc in CA has written about Patricia Kane in his book. I live in the sticks and there are no doctors that would do this on their own so hoping that the doctor I picked will partner with us. Thanks for all you do. I believe you are leading me to get my life back. Before you came along, I was sinking faster than I could swim and I am a good swimmer.
Ya, it seems that fat in the mid section is not so ‘estrogen’ dominant, whereas fat on thighs, breasts and hips is more mediated by estrogen (and low testosterone, but that seems self evident)! Boo estrogen! Makes you cold and fat. No thanks. Luckily CT gets rid of it.
DR. Kruse,
My husband added PQQ to his program about three weeks ago and has been experiencing pretty uncomfortable GI problems. Any ideas why this might occur from this supplement? He is only taking have the dosage. Thanks.
@Susan not from PQQ…..i have never had anyone have that
Couple of questions:
1. Have you noticed any racial differences in how people adapt or process when going “into” CT? (I’m assuming Black folks have some issues due to Na/K channel differences.)
2. Is your wife still body-clipping and blanketing her horse(s) in the wintertime?
3. It looks as though you’ve *lost a little gray…am I right?
@Marie so far no racial differences…….and my wife babies the horse…..but lets his coat grow out in winter. My hair has changed a bit but not much. I think those are CT effects
Dr. Kruse – I have a friend very much in need of your help/diet/advice. He is at least 100 lbs to lose, but says anytime he makes a big change in his diet/exercise he is debilitated by gout. Any thoughts on how LeptinRX/cold could work for a gout sufferer?
Are you more susceptible to fat gain when you are cold adapting? Is that why you need to be ketogenic and restrict carbs? I am already paleo and keto for 2 years. My carbs are often 10-15g per day tops. But say I ate some more carbs one day and fell out of ketosis, do I really need to worry about fat gain?
Also, my appetite is killed doing CT now (good sign) but I am still eating breakfast, although not a BAB, it is a larger breakfast with 2 eggs and some grass-fed beef (small amount like 1-2 oz.), butter added, sometimes coconut oil and raw cacao with 2 drops of stevia for dessert? Is it ok to eat to hunger? So, if I’m hungry I eat breakfast, but if I wake up not hungry, should I wait or eat anyway within the hour as per Leptin reset rules? It feels strange sometimes to eat and not pay attention to hunger cues. Are we supposed to marry Leptin reset with CT?
@CW no I dont worry about Fat gain unless I am creating a lot of mismatches…….the appetite is a great sign and you cant eat a BAB when your cold adapted. I do not do Stevia at all when cold adapting…….in fact I do no sweeteners…….in cold. If youre hungry eat. The Leptin reset plus cold is meant for the obese of those with gastric bypass or those with serious leaky gut issues……..If you do not have those I think CT alone with the keto-seafood paleo diet is all one needs. Seaweed is about the one staple when I am going deep CT. I have not done deep CT in quite sometime…….But next winter I am planning on it for the entire winter.
@Dr. K,
Another video of Wim Hof. Neolithic mind sabotaging our paleolithic genes.
Endless summer inside your cloths. As if.
http://www.youtube.com/watch?v=mE_dFEEYAsA
I love you too Dr K. In a manly way of course. No hugs. A firm hand shake kinda of way .. 😀
@Cooling……..I am just explaining to Wim and the world that this is not magic power……..it is all by evolutionary design.
Dr. Jack,
Have had chronic RA for 35+ years, lots of damage, etc. going paleo has stopped all inflammation but I have questions re how fast I should go with the CT since I just read your comment about those with FM and Lyme. I have joint replacements in 3 major joints too, is icing those joints for long periods of time ok? Lung damage as well with elevated resting heart rate when 02 level drops for long periods, can CT be harmful with that?
Also, what supplements would you recommend for someone like me?
What blood work would be best to start with, am seeing my ob/gyn next month.
I cannot begin to tell you what a life changer this has been for me. thanking you seems so small for such an amazing thing you are doing for people, but huge thank you nonetheless.
I have been going slowly with outside exposure, car AC full blast, cold showers only and spot ice pads on various body parts. In summer will trek to the beach for long swims in the pacific.
@Cecile I think your O2 issue has to be carefully navigated…..i personally think you should talk it over with your pulmonary doc……but I am afraid if you do they will tell you to stop because quite frankly they dont know squat about this…….I am going to suggest that you continue on albeit more slowly than most for adaptation. If you go slower, it will be hormetic and it will strengthen your immune system…….I would tell you to eat Terry Wahl’s version of the diet and marry it to a heavy seafood protein load……in other worrds create your own personal version of the paleo diet that best helps your disease…….AI and cold is a perfect match but while Terry is a big veggie person…….if you do this you must have more seafood and some nuts.
Hi Jack, you say to stick with the LR until all Neolithic diseases are gone. So does that mean it can be stuck to for months to a year? Any harm in that? I think it will take that long for my gut to heal. I am doing CT too. Thank you. Loved the book recommendation and looking forward to your TED talk.
@Natalia I been in for the better part of 6 years…….how is that for adaptation!
@Jack – I’ve seen a number of posts from @Susan, @Mark and others regarding numbness in the extremities during CT. I have the same issue and my feet become both numb and painful, so much so I need to pop my toes above the surface of the water. HOWEVER … whenever I pour cold water over my face and head, my feet and entire body almost immediately feel warmer. How is this possible?
It seems to fly in the face of what I read about the Dive Reflex in mammals, where cold to the face triggers slowed heart rate and vasoconstriction in the limbs. But when I pour water on my face, I feel MUCH better in through the duration of my CT sessions. Am I doing something wrong?
@The Kid…..it is possible because the nerve that controls your facial sensation (trigemenial nerve) connects to the surface cold receptor system…….and it connects to directly to the hypothalamus that controls TRH…….TRH upregulates fat burning at UCP right away when your cold adapted. That wiring is all in CT-6 but it is dense neural science. No way for me to make less complex……..science sometimes gets tough if you really want to understand it……and I always do.
@Dr Kruse:
My wife just spoke to my PCP (who is also a neurologist), and he was quite impressed with the lag numbers improving over 3 months. She told him about CT plus LRx and he wanted your information…was especially interested in the CT.
@BenG anyone who works with the brain knows the power of cold…….it is the rest of the world and medicine who have yet to discover what neurology and neurosurgery knows. I guess my blog is out there lighting the way in this endeavor.
oops..lab numbers (not lag) 🙂
@Jack It is becoming increasingly clear what Factor X is.. 😉
When is the membership site coming online.?
@Resurg……i figured it would…….just keep it quiet if you will until the book. Then I will show you how I got there. I have been leading you there for months…….so I guess my plan is working.
Bringing biology back to its foundational epistemology.
Hi Dr Kruse,
I have been on lowish carb Paleo Robb Wolf/ Sission for 14 months 225lb->182lb . I may have 5 lbs of body fat to lose if that much. (my wife thinks I look good already and thinks any more fat loss will make me look bad, but I want better body composition. ) I do 30 min Heavy weights once per week using Body by Science protocols, but add in a few more exercises. I have done them fasted, but now will follow your protocol. The only thing I have is major sleep issues – I wake up 3-4am and can’t go back to sleep. but feel very rested on 5-6 hrs sleep. but I know I need to sleep more. I bet my cortisone is messed up. may have it tested. I ate only dinner following my hunger cues. I began Leptin Reset 6 days ago and lost 2 lbs. I eat a BAB although not hungry. I began CT yesterday and plan on following your protocol to help me shed the remaining fat, especially spot cold in one problem area and to improve my sleep- which is the main driving factor.
Q: 1. Not sure if I am leptin resistant, without labs, is it OK to do it for 8 weeks anyway?
2. can I continue weight training 1 per week during leptin Reset?
Thank you in advance and for all you do!
@PaleoDentist Youre not LR……CT will shred your remaining fat
Dr. Kruse,
I was wondering if you had an estimated date of when you would be releasing your post on gallbladder issues? I mentioned that most women in my family have had theirs removed and I’m hoping to keep mine a few wks ago. You said possibly after Paleo FX.. so I’m just wondering if you were putting it out soon. LR + CT is going great so far. Thank you!
@Erica Its coming soon after the CT series……
OH, I supplement with 1000 fish oil, 2000 fit D3, Selenium, and Magnesium citrate.
@Jack
1. So ready for the membership site. ETA?
2. There seems to be a little practical confusion on the benefits of continuing one type of CT after transitioning to another. I did my first baths yesterday, loved it for simplicity and quickness to get the most amount of skin down in temp. If you are going to CT for 1 hr a day, is going bath only ideal – or is there a compelling reason to keep face dunking or spot icing. For me, I love the face dunking – but want to get the most bang for my hourly buck.
Thanks for everything. Good luck at TedX!
@Cavemam some snags this weekend……technology issues but its coming in a few days…..not sure if they are working today. CT works no matter how long you do it…….
Q: 1.any harm in following leptin reset for weeks anyway? I eat BAB and DInner only except for weekends where I may eat lunch with my family. How can you tell I am not leptin reset? I went off eating plan for 7 days on a cruise and gained 7 lbs!
Q2: does this mean anything?
@Paleodentist…….its a waste of time for you…….you need to progress on to this……https://jackkruse.com/so-you-completed-the-leptin-rx-what-is-next/
It never ceases to amaze me how humans are fine wasting time…………if it fits their neolithic thoughts or beliefs……but guess what? Time is the most valuable asset we have. If you we use it wisely we maybe able to create more time for us……..by living in that Ancient Pathway as much as we can………..Chew on that. That is how I roll and how I think.
Read What Dean Dwyer said about me today………I think a lot differently than anyone in this space for a reason. The science has brought me to a new place of understanding. Evolutionary biology is the epistemologic cornerstone of medicine for me……..now. Physics and Chemistry has no foundation in science until 1905 when Einstein found relativity and invented quantum mechanics. 125 years ago we have no way to know how to describe Brownian movement, but we knew it was possible. Today, we all know what Armstrong, Phelps, astronauts and Sherpa’s can do is nothing short of remarkable. What Wim Hof can do is amazing. I think I found out why it is possible. The answer lies in our evolutionary history. And I am slowly unveiling it in 2012 for you to digest piece by piece. If I just blurted it out it would stun you and you would race past by it and never pay heed to it because it is that stunning and easy for you to dismiss. When Einstein said that light bends in space for 11 years people mocked him until some scientist went to Africa and studies the perihelion of mercury…….in 1916 and found out Einstein was indeed correct…….and he did it without any testing……..there is now way to enter space in 1905 and prove it………but guess what…….he was dead right. His science predicted black holes and quasars…….it took until 1995 to prove he was right. Todays world requires execution of an idea……..well, that is what I am doing.
When I get done with this…………….it will be as plain as the nose on your face. I am not going to wait 11 years to make our species better. You all will have the choice how you want to live once you see the magnitude of what the Ancient Pathway hold for every mammal on this planet. Our species of mammals has evolved away from this pathway…….and we remain blind to it……..but that does not mean it does not exist………I am forcing you to consider that it is present in every one of our brain’s if we learn how to access it………..and when you realize that I am correct. Well, we all get better for it.
In 1905-1916 we had no way to figure out how to make a satellite cell phone without that data. We had no way to know how to get to the moon. From Einstein’s unconventional thoughts…… science changed in a decade and changed what was really possible for us in the 20th century. See Atomic power, the bomb, and space travel. Prior to him we were all limited by beliefs. See how religious beliefs kept us in the dark for thousands of years. That is a stark wake up call. Today modern healthcare has replaced organized religion as that blinder of truth. I’m shining light under that rock today. In medicine, and specifically biology, for the last 300 years we are also still int he dark that physics and chemistry were in up until 1905 too.
Today, we rely on research literature that is horribly flawed and we think modern RCT’s are “the gold standard” for truth finding. That is pure folly. I rely on the RCT that evolution has provided for the last 4.5 billion years. Its a far deeper experiment in my view. Since I have done this…….I can now explain many things that modern men of science find vexing. They find it vexing because they are caught looking at the problem from an older more accepted view point. I rejected that view point after I read the Monk who sold his Ferrari……..I know what it means to look at life from a new totally outside the box perspective.
But I test this on me………and the results of my personal thought experiments……..have been nothing short of stunning for me and my family and patients. I am about the only person who thinks this way in everything I do now. My life, my emotions, my thinking, my professional life, and the way I live. I live 100% in this pathway now……..for the last 6.5 years. I will never go back. The science is that compelling. The next decision is yours……..and that of the public. I made my choice 6.5 years ago when I stumbled into this science.
http://www.beingprimal.com/21-astonishing-things-from-paleo-fx-that-you-probably-didnt-know
Have a question re: BAB. So you said if one doesn’t have major LR/gut issues etc. then you don’t need the Reset w/ CT, just CT + keto seafood. I pretty much do the CT + keto seafood and not Reset, but what about breakfast? Is it still a good idea to eat a breakfast (but smaller than BAB) since you mentioned that protein in the morning is best in morning and not too much fat (I figure in the morning) in order to avoid insulin resistance.
What is best? Thanks.
@Gladina read what I just wrote to the Paleodentist. If that does not answer it nothing on my site ever will.
p.s. I know that high fat is good, but I am assuming you meant not too much fat in the morning and/or make sure ratio of protein is high enough FOR MORNING so as to avoid insulin resistance. But seems like the rest of the day it’s OK to have higher fat content and veg .
Hey Jack –
I’m sorry if you already covered this, but I recently took a job where I’ll have to wake up at 3:45 AM and be at work by 4:45. I’ve been doing the leptin reset for a couple months now, and I usually wake up naturally right at 6AM. Obviously, this job is going to throw a wrench into what I’ve been doing, but, a job is a job. If I can manage to stay asleep from 12-3 and continue the reset accordingly – what are the implications of eating breakfast by 4 instead of 6? I’m not leptin sensitive yet, but with the new schedule, would it be better to wait until 6 to eat my BAB rather than eat it around 3:45/4? Am I even asking the right questions?
I’m kind of really upset that I can’t work my way to optimal with this new schedule, but I’m just trying to figure out how to make the best of a bad situation.
Thanks for all you do.
@Lindsey you just took a step into sub optimal……..shift work will kill you faster than you can imagine. Your choice of what you do……..might choice should be intuitive to you.
Jack,
I just watched a video of Wim Hof being injected with dead bacteria. Two observations
1. His blood pressure looked high on the ekg 165/83, in room temperature (on a hospital gurney) I would think someone in such good shape would have a low blood pressure. Maybe the anxiety of the test?
2. After successfully completing the test, the doctors said his cortisol levels were much higher than anyone else, which keep his immune system at a very high level. I thought high cortisol was related to all neolithic diseases and is to be avoided at all costs? How is he able to deal with the high cortisol without the deleterious effects on his body?
The Best
@Conan sustained cortisol is……Wim is cold adapted so he can raise his when he needs to…..and then it drops. We are built for short duration insults……not the ones modern life inflicts upon our physiology.
My CT update:
I’ve been doing it about a month (and doing Leptin Rx about 25 days) and I’m having really good body comp changes!
I don’t really need to lose weight (I’m skinny fat) and I’ve only lost 3 lbs, but I’ve lost 1.5″ around my waist/muffin top. I can tell I’m getting a lot leaner. I love that you really can spot reduce. This way I won’t get too skinny everywhere else. My hormones are really changing, too. I had my first non-excruciatingly painful cycle and my cycle length has gone from being too long to too short (right after I started CT) and now closer to ideal.
Other CT benefits I’ve noticed: softer skin, I get sleepier in the evenings, better singing voice (must be less inflammation in my head and vocal chords) and my hair is very recently starting to feel thicker. This didn’t happen when I did Leptin Rx before. I can also do more pushups and kettlebell swings;-)
Ice is magic!
My hubby is LR’ing and just started CTing yesterday and he’s lost 8 lbs in his first week!
@Erin Awesomeness……….!
Good morning Dr Kruse,
I came back from an easy walk to feed the cows and my temperature is 35.2C(95.4F). Could you explain why a lower body temperature is better than normal body temperature of 37.0C(98.6F)? How can one radiate heat from skin (be warm to the touch) while having a low core temperature? How does this work into CT?
Cheers,
Caroline
@Caroline……as on radiate heat from BAT the heat leaves the body with the fat source which is the fuel…….what is left behind is lower levels of core heat. Remember the fat is only designed to have to last 4-6 months while fall and winter lasts……but for some modern humans they have years of fat upon them……..and they have trashed their physiology over that time……so adaptation may take several years to get back to normal…….my body temps took close to 3.5 yrs to normalize……but they come back to allostasis eventually.
Oh, another interesting benefit: I’m not sure if this is from CT or sublingual B12 (or both!) but my face/lips have more color. I look a lot more alive now, lol.
@Erin it is because you are more alive. See the longevity angle. Many people make comments about skin color and CT……See Sherpa’s and Inuit’s…….alpha MSH upregulates melanocytes because it reduces the chance we get frost bite from the cold…….it is also why polar mammals are all white and black in some fashion……..evolution has a plan. We need to follow that plan.
K Dr. K. (lol, that’s supposed to be funny). Will be more Post Rx with the increased seafood + CT.
😀
I sometimes feel like this journey is long and it can feel discouraging…BUT, if I recall my sudden jump in performance in karate last year…it was two things: 1. I was eating paleo (80/20 ala Mark Sisson), but not only that, it’s b/c the way the body gets adapted. Like it’s a distinct JUMP. Of course I got myself in a hole in the last 7 months which I’ve been working on to correct over the last month. So I remind myself that it might still take time. Once I get to Optimal—I’M NEVER LETTING GO AGAIN!! I can’t go through that AGAIN! No thanks! Once hormones get screwed, it’s HARD to get back…lesson learned.
@Gladina……..is evolution easy? Look at the modern day polar bear? Or how about a sabertooth tiger? A wooly mammoth? Life is not meant to be easy…….modern humans forget this because our brains make life easy…….and when it becomes easy we begin to die slowly because of it………my job is to remind you of it. Wild animals do not get to eat 80/20 I reject that nonsense too……….This is not meant to be funny……there is a deep lesson here.
@Erin, funny that you talk about the color of your face because that is one of the first things I noticed when I began to CT! Other people noticed it as well & when I tell them what I am doing they give me that look like I am crazy. I figure soon enough they will be back for more insight. Like Dr. Jack says, I am fixing myself first & will be ready to help others when they are ready to fix themselves. Loving life is a blast!
@ Erin. My cycle is still stupidly long. Can’t wait for the day for it to be NORMAL length. (Again, last summer when I was more optimal it actually was 21 days ‘twice in July’. It was easy and painless. Want that back).
Waiting…
Your response came first. Yes, that’s why I’ve started over the last month w/ CT and keto seafood. Remember I am the one that has said about improved eye muscle. So I KNOW it’s working. I’ve also reduced in swelling on my lower body. Also my skin is darker too as mentioned above from Erica and Larry.
Another thing, with the little heat wave we had I got super tanned so easily and didn’t get the ‘first burn’ from lack of exposure all winter. Even though I’m olive complexion I still had that problem on the first time I got tanned in summer. Had two other friends have the turn red but turns into tan later. I know I’m healthier for doing this.
Yes, like Larry, I’m trying to keep my mouth shut on my beneficial changes (unless if directly asked…)When I have more full out results then I can be more forthcoming.
Alright Doc, I gotta say…this CT stuff is kinda crazy. Now I’m not real old (33) or really overweight or out of shape. But I’ll tell you what, I’ve been averaging 3 I’ve baths a week the past month, coupled with cold showers, and while it seems I continue to lose more flab, there is one thing I REALLY notice (and I’m not trying to be crude or funny).
My sex drive is insatiable. I’m driving my wife nuts, cause I have the sex drive I had when I was 18. Waking up at full staff, being turned on constantly, this is something else. I’m not sure if it’s a completely good thing though, cause I’ve been grumpy when the wife doesn’t cooperate (if you know what I mean). But my wife even said whoever came up with cold showers calming men down is an idiot! I feel like you’ve helped me turn back the clock 15 years!
@James…….This is why in CT-6 I made light of Kresser and Masterjohn’s podcast on eating Moose thyroid for reproduction. It is total BS……..if your cold adapted. If your warm adapted you might need some moose thyroid. This is why they both believe this stuff. They have no clue that there is a wild optimal side to our biology. LOL If you ever meet my wife ask her what cold really does……when you husband spends 6.5 yrs in the ice………just ask her. You might just have to look at her face and she might not have to say a word.
CT is just pure ridiculous for our species and the quicker we all get to it……..the healthier we all become, the more disease we can cure, the longer we all live and our species will begin to look like what Michaeloangelo carved into that White Carrera marble……….that was my spark of intuition lighting my candle………..
@James Duffy…I can also say, the morning wood has made a full recovery.
@BenG awesome stuff BenG! Ice is cheaper than Viagra!
@Dr Kruse. Cold kills fat cells. Dieting shrinks fat cells. Dead fat cells release the toxins that shrunken fat cells from dieting doesn’t right? Also, why does the literature say fatty liver get worse on rapid weight loss?
@BenG because they dont lose weight the way mammals are supposed to………in the cold of autumn and winter in the poles or in the dry season in the tropics when little food is around.
This is why is stays oin our blind spot. We no longer face a true winter any longer as humans…….we don’t hibernate, we wear clothes, have heated escalade seats, fur coats, heated houses and eat banana’s year round while we exercise our selves to death…………because we are unaware that the biochemistry of cold is built into us to balance the warm pathways of biochemistry.
Excellent publish, very informative. I’m wondering why the opposite experts of this sector do not understand this. You should continue your writing. I am sure, you have a huge readers’ base already!|What’s Happening i am new to this, I stumbled upon this I have found It absolutely helpful and it has helped me out loads. I hope to give a contribution & aid different users like its aided me. Great job.
Hi Dr. Jack,
The CT adaptation is going pretty well. The detox reactions are less severe…no more headache/nausea/muscle soreness. However, I’m more hungry, not less! Really struggling here. Had to go back to eating BAB and lunch. I’m eating more, but staying ketogenic. What’s going on? I’m taking a 55 degree bath for 1 hour daily. Just hit the 3 week mark. No weight loss either, but of course that could be because I’m eating more.
@Kathylu if more hungry you are not finsihed detoxing……cold destroys hunger. it is impossible to be hungry in cold……so if it persists that means you need work up. My bet is something else is cooking
T Van Djik — your gf might have an enzyme defect called MTHFR. It can cause depression and treatment is either high dose supplemental folic acid (prob not a good idea) or taking the active form of folate L-methyl folate. High homocysteine can be an indicator, otherwise can do genetic testing for MTHFR. Lots of info out there if you google it.
Is it ok to stay in the cold pathway of eating during the summer? Will keeping carbs to a minimum create more mismatches or is keto ok all summer long as CT is involved?
@jeremiah yes it is.
I’m sorry to ruin the party, but apart from feeling more upbeat, and worse sleep (yes, worse, and usually I sleep quite well) nothing impressive to report. The little portion of belly fat that I have, which neither paleo nor HIT nor IF have removed, so far CT has not helped much either.
Doing 20m cold (13-14ºC) showers per day+ 3 extra cold showers in the evening per week.
Best.
@Santiago…..no party to ruin.
I haven’t lost any weight from CT yet, but I think SOMETHING is beginning to finally happen, because it seems like my belly fat is getting much “softer” and squishier. Hopefully, that means a change in composition will soon follow. 🙂
Doc,
Something I’ve noticed lately when CTIng for an extended time, but usually for a while when I get out of the bath, is heartburn. Not too severe, not always, and it subsides, but it has to do with lying in the cold horizontally or so I gather. Paleo Keto food wise, no crap. Any guesses?
@Laney good sign…….it has to do with your gut healing.
hi dr. k,
may i ask what your % body fat is, and if it is not where you want it, why is that? thx
@V I am getting it tested in June. Last June I was at 15% on BIA and 17% on Dexa
Hey Jack, I have a strange (maybe not) observation. I have been on the leptin RX since 3/2 I’m down 20 lbs, started CT a week after. I cant workout due to Shoulder and clavicle dislocation. Now I have noticed my muscles getting bigger and harder (arms,calf’s & unmentionables) Am I delusional or have you seen this already.
@Mike this is precisely how CT works. It increases your Testosterone and GH without any exercise. People like Ken O’Neil do not think this happens…….it works in polar bears and it works in us because this Ancient pathway is built into every mammal on this planet. Awesome stuff.
Hey everyone, I just wanted to relay a fascinating real-life story of how amazing CT is. So my brother’s friend’s wife went in for a c-section, but unfortunately the surgeon accidentally cut the placenta, which means that the baby doesn’t have access to oxygen anymore. So they got the baby out as quickly as they could and gave her three epinephrine shots (I guess that amount is unheard of) and then as a last effort they put her on ice. Long story short, 72 hours later she started breathing on her own and just 13 days after birth, she left the hospital with full motor skills and is even breastfeeding. The MRI does show some evidence of brain damage, but they are not seeing any outward signs of it. Unbelievable considering that they initially sat down my brothers friend to talk to him about planning a funeral! This made me think about Kevin Everett, who was paralyzed in football, but regained movement as the doctor used cold therapy on the way to the hospital (http://www.nytimes.com/2007/09/16/sports/football/16everett.html?_r=1). Anyways, just another example of how amazing this stuff is. Thanks for putting it out there for all to see Dr. Kruse!
@Mark awesome stuff Mark and thanks for sharing this with people. Kevin Everett is in my book and he is part of the reason of how I found out about Factor X……….ironically.
Take a look at this link sent to me today by Kevin C…….from 1993 before leptin was even discovered.
http://www.me-cvs.nl/index.php?pageid=3076
In addition, I also just wanted to write about my progress with CT. It definitely has not been a straight path, but I’ve made progress nonetheless. Yesterday was my best ice bath yet. Stayed in for 70 minutes and could have stayed in longer. Water started out at about 53F and finished at about 63F. Feet were submerged the whole time and didn’t get white or numb this time (I found that stretching my foot by straightening my leg every so often really helped). Hands were even submerged for the last 20 minutes. No knit hat anymore. Shivering was pretty steady throughout, but bearable. I had about 4 pounds of ice on my torso. After I got I out, I felt fine. Very little shivering and warmed up fairly quickly just doing stuff around the house. The pink skin that had the ice on it returned to normal color pretty quickly too. Overall it was a good experience, but the point I want to make here is that it took me awhile to get to this point and while I still have further to go, I now feel confident that I can do this and won’t give up. For example, I found what works really well for me is to start off by taking a cold shower for a few minutes at about 65F. Then once I’m used to that, it’s almost easy to turn the temp down as low as it will go (usually somewhere between 50-55F at my house). And then doing the tub is no big deal because I’ve already gone past the hardest part. Anyways, hopefully this will help someone push through and keep trying to do CT. Find a way to fit it in, even if that’s ‘just’ a cold shower and not a bath (the time for a bath isn’t always there), at least that still something. You probably won’t be perfect everyday, but keep on trying.
Dr. Kruse,
You and I discussed some of this before in other contexts, i.e. I have trauma induced spine problems, epilepsy, osteoporosis due to long-term phenobarbital therapy. Recently, I fell down some stairs as the result of a seizure and injured my neck. The diagnosis is multi-level (C4-5-6) cervical stenosis with myelopathy and my physicians are recommending a horrible sounding procedure called Cervical Corpectomy and Strut Graft. As a neurosurgeon, do you think CT could play a part in my recovery and might I be able to avoid surgery?
Thanks.
@Paul I looked at your case and I sent you back what I thought. I do not think you need a corpectomy……i think there is a better option and yes I would use CT on you both pre op and post op. You could do a trial of CT to try to avoid surgery and see how it goes……but with a seizure history you must have a spotter when you do it……if you seize in the tub you could drown. Not good. So make sure you take appropriate actions after you decide what to do.
I am doing CT to help with wacky hormones and have confidence it will work but also have a stubborn case of vulvodynia. That was brought on, I believe, from repeated bouts with Candida (a result of a SAD diet for so many years). After various unsuccessful treatments, my OB/GYM stated there might be nerve issues and prescribed Gabapentin (which I chose not to take). Any thoughts on whether CT could resolve my problem? Maybe add epsom salts to my cold baths?
@LD i have one patient right now with your condition and she is using this……no results to report yet. Keep me in the loop on your results. This is a very tough clinical problem to solve for women.
Paul, good luck to you!!
Mark, thanks for the advice.
From the article about the football player:
“But studies have not always borne out the initial promise. For example, although neurosurgeons had high hopes for hypothermia to quell damage from brain injuries, a rigorous study showed it did not help. In fact, patients who were chilled actually spent more days in the hospital with medical complications than those whose bodies were kept at a normal temperature.”
dr. k, any comments on the above? actually, i was thinking before i read about the football player about if it would be possible to put the very ill in a medically induced coma and do CT. just thinking off the top of my head.
dr. k, to do you a goal for yourself for % bf? what has informed that goal?
thx as always.
2V the General surgeon I presented at Paleo fx with spinal cord compression and quadraparesis is not back to work and fully into CT…….his dad was neurosurgeon at Vanderbilt……..and even his Dad was shocked the the results………The way I am applying the CT is way different than the studies. Why? I found the Ancient pathway in the brain and worked out how it works in us. The results will posted in the comments of the blog I would imagine for a long time. There has been a lot of magic coming out of my clinic here in Nashville. People can apply this at home without any special equipment to help themselves tremendously using the cold with a cold adapted paleolithic diet.
Just curious. What, if anything, does sound/noise have to do with being Optimal? It seems like nearly all the senses have been discussed, but I’m wondering if the noise levels we are exposed to in the modern world have an impact on us? I know it can cause stress..which raises cortisol..
@paleoNana ………cold dark and quiet is a great way to live.
Doc,
Thanks for the response in #286 as regards heartburn after extended CT’ing.
Another question: You mention sleeping ‘above cover’ with little or no clothing, in the dark. What is the ambient temperature for this (say as expressed by what you do for yourself) and at which stage in CT would you recommend giving this a try?
@Laney in my house my wife can tolerate 60 I would go to 40 if she’d let me. I love the cold.
Dr. Kruse, this is amazing! not only do you provide all this info but answer everyone’s questions. Thank You!
@paulalynn I try…….
Dr. K – Re Comment #292 – Ken O’Neill probably needs to take a visit out to Vasper (company has NASA ties) at Moffett Field in CA. Seems they believe in the Growth Hormone and related effects in T based on CT. http://espn.go.com/espnw/training/workouts/6254237/my-revolutionary-nasa-workout
Even Tim Ferris and other Olympic athletes have been there to check out the workouts and CT. http://www.vasper.com/index.php?option=com_content&view=article&id=31&Itemid=105
Might help his ‘think tank’ think out of the box down in Wimberely a little better.
@Kevin when people are steeped in dogma it is hard to change. I know……my job is quite dogmatic. If I could change my thinking anyone can. Using evolutionary biology as my North Star made it easy.
@Kami-I used to keep my feet out of the water with socks on and then started putting them in with my Neoprene socks. I am also finding it harder to stay in the water now that I am using the socks. How are your feet in the water now? Are you still doing okay? My feet hurt so badly in the water that it is tough for me to take. How did you do it?
So, while I am doing CT, I don’t need to be worried that I will gain if I am not perfect with my diet. I am as paleo and keto as I can be, under 15g per day, I eat some salmon, shrimp, several times a week, but I am also eating 99% dark chocolate. And I’m starting another hcg round in April because the weight just won’t budge. My appetite is low and I eat less but that doesn’t seem to cause the weight to drop. It’s hard reading about others who are dropping weight so quickly. Most of them are men I think. Is it ideal to do the CT while taking the hcg or really ramp up the CT when done taking the hcg in the maintenance phase?
@CW depends how bad your diet is…….but cold helps tremendously with weight loss.
Thanks, yes it is a difficult problem and as you can imagine, emotionally very difficult & challenging to my marriage. I try not to kick myself for the years of crappy diet but what’s done is done and now I am working on being optimal.
Doc,
Do you know much about muscular dystrophy? I’m wondering whether CT could have any benefit.
@Rook I know about it but have no one using CT with it. I do not think it would hurt that is for sure. Since CT is ananbolic for muscle I think it would clearly help…..but no way to say for sure.
What can I do for estrogen dominance? Will CT help? Leptin reset? Can I still do hcg? I am up to 30 min. cold bath, added ice last night but water temp still around 56 with ice since it’s coming out warmer now. I am not treating the ED…will test again soon to see how bad it actually is. But I don’t go near soy and haven’t for many years, don’t use plastic, minimize anything like xenoestrogens and phytoestrogens for years. I don’t know why I even have ED. But I think once you have it, it really hard to fix it. I was on progesterone cream once 5 years ago, but then, I got pregnant and had to discontinue it at some point in the pregnancy. Didn’t start it again after.
@Claudia HCG and estrogen dominance never work well…..most women who are estrogen dominant gain as soon as they come off protocol. The key is CT. HCG works best in the cold for a deep rooted evolutionary reason………..
Dr K, in the past I would typically catch a bad cold after staying outside in the cold without sufficient clothing. Today I walked briskly 25 min to the train station and then back wearing just a short sleeve shirt. It was 40 degrees and windy here in Chicago. No symptoms of cold so far. Are there any safety boundaries to be aware of? (I probably missed this info in one of the earlier CT posts.)
@GeneK using deep CT Wim Hot was able to inject bacteria and not get ill……..I was able to withstand something more radical with no ill effects. So far I know of nothing that CT does to make you ill. It strengthens immunity as I laid out in CT 6.
Jack,
I think a lot of us out here would find it very interesting about a typical week of Dr. Jack Kruse. IE: breakfast, lunch and dinner meals, workouts you do, how you do CT and when, supplements you take and when, snacks if any, occasional cigar? Or cocktail?
The Best
@Conan this year has not been typical for me…….after 3/31 you’ll know why. I may get a blog into my normal routine later in 2012 if people really want it. I am kind of boring. Cold dark and sleepy.
Dr.Kruse,
I have just read ( on Animal Pharm )an alarming find on seafood both farmed & wild, & pesticides & how they are the most toxic0 :- Fish: One of the Most Pesticide-Ridden and Toxic Foods
My family and stopped eliminated 80-90% of our fish and seafood consumption (both farmed and wild) when I was first pregnant 12 years ago. I figured what wasn’t safe for my baby and I just was not safe, PERIOD. We try to eat ancestrally but seafood just is not part of the equation at this time. Many cultures who subsist on fish and their marine predators (seal, whale) are documented to have elevated levels of persistent organic pollutants (POPs, pesticides, PCBs, solvents, etc) and heavy metals: Great Lake Anishinaabe, Arctic and Greenland Inuit, natives of the Alaskan Aleutian Islands, Amazon Brazilians, Peruvians, and Faroe Islands inhabitants. Where is the source? Pesticides and industrial pollution are bioaccumulated in algae, daphnia, marine life and large predacious fish and marine mammals. Human variance shows that not everyone is severely affected by heavy metals (mercury) and pesticides but certainly some are more sensitive than others or bioaccumulate at extremely higher rates than other individuals. The carriers of apo E4 allele, the ancestral ‘efficiency’ allele, appear to exhibit higher harboring and decreased detoxification of trace heavy metals (iron, copper, lead, mercury). This may explain the link between increased incidence of central obesity, metabolic syndrome, T2DM, Alzheimer’s and dementia and those of ethnic descent where the apo E4 allele is more dominant (Inuit, Amerindians, aborigine subpopulations, northern Chinese, northern European, Africa).
Do you have any comments re. your fish protocol?
@Paul Grace is a rockstar and she is very concerned with heavy metal toxicity…….I think she is right about many fish having issues but her defense is to eliminate fish……..my defense is to make sure my immune system is so good that the toxins never cause me an issue. CT does just that. See when you have a bad thyroid, LR, terrible adrenal gland……..well………you have to eliminate things to stay healthy…….but the tradeoff is that you cant have the most nutrient dense diet that the Ancient Pathway needs to run optimally……..if you at CT and cold……well the fish issue goes away. How do I know this…….i test myself for this very issue every three months………i believe in a quantified self testing platform. I leave nothing to chance. I question everything.
Dr. Kruse,
Question: I don’t care that much for longevity I just like to live my life the way I like it. At the moment I’m on keto diet and don’t intend to eat carbs ever again (except some occasional small amounts here and there, I just don’t like them on a daily basis) .
I don’t get too much sleep, sometimes I’m up the whole night. I like endurance training.
Is any sense for me to do your CT protocol. I don’t mind feeling cold I used to bike/run during winters with no problem.
In other words if I’m not going to implement your other recommendations (early to bad, no lights after sunrise, easy on exercises, carbs in summer, etc) does it matter if I’m cold adapted or not.
I may just do CT for fun since it sounds like fun but I just want to know if it will bring any benefits.
Is the protocol form Becoming the Iceman effective to be cold adapted?
Regards,
Paul
@Paul since you like things that are biologic opposites…….yes CT can help…….endurance exercise destroys sleep and CT helps sleep…….so if you want to keep doing endurance exercise you must sleep…….so make sure you employ CT. you wont be healthy or live long but that is your personal tradeoff and you said you are fine with it……..be safe friend.
@Dr. Kruse -I just wanted to run this by you to see if this is an example of ‘detoxing’? So since Sunday, the last two nights I have slept very heavily and have been unable to get up when I want to (400). Instead I have been dragging myself out of bed around 500 with the feeling that I could sleep a bunch more. Last night I was even asleep by 900. Yesterday I was also in kind of a fog at work: couldn’t concentrate that much and little motivation. And lastly I’ve been getting some nasal discharge like I have an infection or something (sorry to be gross). Does this mean that I have to up my B’s and Betaine HCL’s to help ‘methalate’ all these toxins? If it matters, I have running my head under cold water before bed to get in some CT since I only have time for a quick cold shower in the morning instead of a bath. It’s probably just all part of the process but just thought I’d pass it along to you. Thanks
@Mark nope that is the CT affect on sleep via increasing DHEA and lowering of IL6. Your hypothalamus is rewiring……..all good stuff.
@Dr. Kruse – Great! I’ll fit in the longer baths on the weekends when I have more time and just get more sleep during the week. Head dips and cold showers during the week, long baths on the weekends, that sounds like a good plan.
I thought that I did look a little leaner today too. Thanks Dr. Kruse!
@Dr. K I found a 1000mg fish oil capsules of which there is 300mg Omega 3. These are cheap and made locally here in Egypt.
But, they have an added 100mb wheat germ oil.
1- You mentioned “reversing IR one would need 3-4g of O3 daily” Does that mean I need 10 capsules to reach the needed ammount? or 3-4 capsules?
2- do you mind the wheat germ addition they put in it?
🙂
Cooling
@Cooling no wheat germ……at all. Your better off with Krill oil 500 mgs 3 times a day. buy it off the internet!
Dr. K, is it OK to walk 2 hrs a day? Sometimes it’s necessary to walk that much in order to get to a destination I need to get to. I feel like CT makes this walking feel like a 15 minute walk instead. There’s no pressure on my feet or legs at all.
Maybe I’m still having some subtle detox symptoms still…is it possible for CT to delay my cycle SIGNIFICANTLY? As in ‘reset’ it? I’m still waiting 36 days later… I just know of this other commenter who said it made hers come exceedingly sooner. Everyone is different I guess. (I’ve had it come two weeks late even after a 30 ish day cycle before, so I probably shouldn’t be so concerned.) But last July when I was going way more healthy, it actually came after 21 days (so twice in the same month). I felt so healthy and good.
I want to ask more questions…
p.s. I’ve switched to the Post Rx and using keto seafood + CT.
Dr. Kruse,
It has been a Month now of long cold showers from our 55F well water. I get cherry pink and numb to the water on my torso and skin temp gets down to 65F normally. I thought I was getting the most I could from a cold shower.
Yesterday I read the 1993 article about Professor Kakker and his work with cold therapy. He suggested that the need for turning on the cold sensitive areas of the skin will prepare the brain for the signals it is about to receive. The procedure calls for a few minutes of chilling the feet first, followed by the legs. The third phase is then the full torso including the neck and back of the head and facial area.
I followed this procedure last night and made sure I stayed in to get the full benefit of the shower. I have to say I got the best CT session I have had and for the first time experienced the “After Drop” of feeling very cold a few minutes after drying off. I felt chilled for an hour after and when I went to bed I was still cold for some time. Not in a bad way but I knew my pilot light was going full blast. I also gulped a large glass of ice water down to reenforce the effect just before bed. What a great feeling. I’ll definitely do this procedure again as it seemed to prepare my brain for what was coming.
BTW, my blood sugar is now normal, blood sugar is averaging 110 and psoriasis has healed about 80% after 40 years of looking angry. The few weeks of sun we had recently gave me a nice skin color that looks and feels healthy. I feel great! Thank you for sending me down this path Dr. Kruse.
@Eric great for you and glad you have persisted. The Ancient Pathway is nothing short of amazing for humans.
Sorry, I meant to say my Blood Pressure is now normal 124/62.
Dr. Kruse,
There are several people named “Paul” on here. Are you able to differentiate? I hope so.
@Pauladams I will have to use your email name then
Every reader here will attest that You are far from “Boring,Cold, dark and sleepy” a.k.a comment #325…
you are interesting, warm, bright and awake.. In a very ‘chilled out’ way 🙂
Eagerly awaiting the post 3/31 Dr Kruse..!
Dr. Kruse, question about CT and temporal mandibular joint dysfunction (TMJ):
I’m undergoing orthodontic (phase II) treatment for this after appliance therapy with good success. I still have occasional flare-ups and suffered one this weekend that resulted in a 3 day migraine and ultimately, lidocaine shots in a couple of tendons. But before that, I put cold packs on the upper back of neck, ear, and jawline for the 1st time ever and while it provided temp relief, the pain came back with a vengeance. TMJ specialist Dr. Simmons said cold ONLY for muscle spasms, not tendon pain/soreness – that it would aggravate them and cause more tension and pain. I assume he’s nowhere near as knowledgeable of CT as you are, but cold did make the pain worse for me once the cold wore off.
Do you have any thoughts on this? I tried to keep cold on the pain spots for over an hour and while it numbed the pain, as soon as the cold wore off, it was worse than ever and causing referred pain in new places. If CT is good for everything, wouldn’t it ‘eventually’ be good for tendon pain too?
Ice internally on the ptyergoid muscles and the temporalis externally……I used to use this all the time when I was an OMFS and treating hundreds of TMJ patients.
Very hungry today. though not as in ‘my stomach is growling’ hunger, but in that I can’t stop thinking about food. Not carbs either, just any kind of food. Weird…
@Mark eat meat…….or offal or fish
Just wondering about your comment “not a fan of flax” Is there any harm tho? I am 48 yr old woman and concerned about phytoestrogens. I make a breakfast muffin with flax, egg and coconut oil. No other carb. Very filling and fast. Love my cold baths and sleeping well. Thanks!
@Bonnie Flax is not a good source of 03’s Krill oil is a way better choice.
Jack said: Ice internally on the ptyergoid muscles and the temporalis externally……I used to use this all the time when I was an OMFS and treating hundreds of TMJ patients.
OMG! A solution to the TMJ I’ve had for 40 years!?!?!
Sally googles the pterygoids…
Do you think I can still be successful with hcg and CT? I started CT several weeks ago. I did the Leptin reset in November through December and post-Leptin after that. Although dairy had been sneaking its way into my diet in the form of full fat greek yogurt, I have eliminated it now. My plan is to start hcg while it’s still early spring, continue CT while doing the hcg, and really ramp up the CT after the round is over. I did gain back the weight from hcg I did last year, but I didn’t gain it right away but very slowly each month I gained 2-3 pounds with my cycle. I was hoping the Leptin reset would correct his but it didn’t. I keep gaining weight each cycle and this last cycle has not been any better, even while doing CT. I can’t lose weight…Leptin reset didn’t work, nothing works for me to lose weight except hcg, and I need to stop gaining, that’s why I feel desperate to start hcg again and quickly while it’s still early spring in NY. Otherwise I would wait for fall/winter.
@Claudia HCG and estrogen dominance never work well…..most women who are estrogen dominant gain as soon as they come off protocol. The key is CT. HCG works best in the cold for a deep rooted evolutionary reason………..
@Dr. Kruse – general question. Since you release fat during CT, should you consciously restrict fat intake since the fat released from the CT could already be put to use? Or is that fat toxic and unable to be used? Or should I just wait until the CT takes over and induces calorie restriction naturally happens?
@Mark you should eat more fat with CT not less.
@Jack What did you do that caused so much of you to be numb for so long? Is it a result of CT?
@Michael It has to do with CT and my ongoing current biohack…….that soon will be public here soon
@Dr. Kruse – Extending on something else, I’m thinking of just fasting while at work going forward and then just have dinner when I get home. My concentration and productivity the past two days have been horrible and fasting usually helps those things. Would this be a bad idea if I’m following the outline that I laid out earlier (cold showers and head dunks during the week, ice baths on the weekend)? I can push past hunger or maybe I’ll just bring some nuts and seeds to work to get me through the tough parts of the day.
@Mark I’d be fine with nuts……but me I would increase my B vitamins and mushrooms at meals and try to avoid snacking. I am no fan of snacking period.
…so essentially I’m just hoping that the CT would trump the usual negative side-effects that one would see with calorie restriction. I think that it would but my mind is so numb right now that I’m not sure. Thanks
@Jack Is it something any of us are likely to run into with the protocol you have laid out, or is it caused by something significantly different?
HI Dr. Kruse, I haven’t slept in days. Still trying to get a doctor to start Phosytidylcholine pushes. Do you think it is the metals being mobilized by CT that is causing me to not sleep?
@Kami I would not test until you dumping is over.
Thanks Dr. Kruse, I need to get back to the basics (meat and fat breakfast, try to IF until dinner, CT with showers during the week and baths on the weekend, keep carbs low) and stop over thinking things. It’s just that these swings in mental focus are driving me nuts. I need to remind myself that there will be good days and there will be bad days on the path to becoming optimal.
@Mark we all need to get back to the basics.
@Dr. Kruse:
I’ve been on reset for about 9 wks. Thinking of moving to post leptin protocol. Have most signs except confused on two.
1. Sweating. What changes in sweating am I looking for? What is “good” or “bad” sweating?
2. Post workout recovery. Assuming one is out of shape and hasn’t exercised in many months. How many days, if at all, should we be sore after a heavy lifting session? What counts as “good”, aka leptin sensitive recovery times?
My last question involves skin. I have Keratosis_pilaris. No one seems to be positive on what causes it.. things from gluten, to dairy, to problems utilizing Vit A are all listed. I have been gluten free completely for a little over 2 months, doing ct, and added liver to my diet. No changes in kp yet.. which is about the only thing that hasn’t changed for the better on your protocol. DO you know what may cause it or if it’s a vitamin deficiency? How can I fix it?
@erica sweating is a warm adapted pathway function……in CT sweating essentially vanishes. Recovery simply becomes amazing with CT. Keratosis pilaris I have no dealt with personally in my practice. My bet is CT plus a paleo diet makes it a non factor like just about any other disease
Dr. K, my 16 yo daughter has been doing the Leptin Rx for almost a month along with CT (not everyday), she isn’t really overweight but wants to get optimal for her sports she’s in. This past week she has been having dizzy spells and blacking out when standing up. We have checked her glucose and it is normal, her blood pressure however today was 95/57. Is this a side effect from the reset or CTing?
@Beth that sounds like an adrenal or aldosterone issue…..if your daughter is overweight by a lot she may not be suitable for the sports she is doing……Her BP is not that low but just have her increase some salt intake and see how she responds. Bacon would be a nice way to solve that.
@Jack,
I have an appt to go try a new doc, and get some of the labs run. I’ve been CTing for 3 weeks now, Typical detox symptoms underway. I’m 10 yrs post bari surg. 2nd time with Reset (this time + CT). Been paleo for 9 months and LC for the most part.
Would the crazy things going on from CT detox make the labs useful right now?
@Cavemam no labs until detox dump is done……i wasted lots of money doing that.
Hi Dr Kruse, a bit off topic, can you please tell us the amount of each ingredient in your optimized coffee. I know it has coconut oil, turmeric, cinnamon, nutmeg, but in what amounts???? Thanks
@paleo dentist It is in the e cookbook.
@Claudia.. Good question!
If CT needs more fat not less & HCG + CT will light the world on fire.. Then the HCG protocol should contain fat… That is where I am headed but (still) all this talk about Sticking to Dr. S no fat protocol has me second guessing myself.. Darn Dogma!
Just recorded a blood sugar below 100 for the first time in a long time. So happy!
@ Sharron Awesome!
Thank you Dr. Kruse, she is maybe 15 lbs over weight tops and is a swimmer. She loves her bacon and salt so we will try to increase that and see if that helps.
bought it!
did 1 hour in the lake today, reading the monk who sold his ferrari – love it
@Julia that book will be the feature of my TED talk this Saturday here in Nashville
thanks for info #364, don’t do labs until toxic dump is over. I can’t even get my lab here in NM to do the correct labs anyway. Example: They did a Vit K-1 instead of Vit K-2, The VAP I asked for came back as a Valpoic Acid the first time, the second time it came back as a Lipoprotein Fractionation panel. I finally googled the proper tests and copied out a sample page for each and will eventually get it to the Dr. and the lab. Right now I have 6 intense weeks of travel and work with HEDIS coming up. Can only do so much.
Question, up till I got so out of shape 5 years ago I used to do a variety of Yoga, Tibetan breathing exercises. I am thinking of executing some of these when I go up to my 10,000 feet playground. I am still adjusting to the altitude so will go slow. Is there value in this or should I just try it and see what happens?
I went up via the tram for the 4th time. I strolled slowly around for the entire hour. At half an hour my HR was 125, at an hour it was 100 and after resting up there for 20 minutes it was down to 78 and at home it was back to 68. This time no hunger but was a little nauseated and light headed but no frank dizziness. I guess I’m adjusting. Right?
Still having a lot of body pain and am doing colder baths in between the altitude. Am I headed in the right direction with this combo altitude and cold baths?
@Terry F keep it up. Your on the road to Optimal now
i really cant wait to hear about that speech. You have been given a gift Dr. Kruse, and you are using it as mothernature would have wanted you to. You’re great!
@Jack: Ok, so since I’m looking for the signs of being LS to change to the post leptin protocol.. I should not be sweating even while working out?
@Erica you will sweat…..but in CT it is rare……because your excess calories are converted to pure heat by thermogenesis and not used to cool off.
Doc this post cleared up many things for me. Absolutely fantastic! I have been doing full submersion in a 51 deg pond for 10 -15 min every day for the past 9 days. This will lower blood sugar as much as 60 points, but it also lowers my oral temp from 95 to 92.4 for 45 min while I shiver then it starts to climb again up to 97+. Is this ok? Otherwise I feel great. Muscle tone is incredible.
Jack, I just got your e-cookbook. I guess the CT helps brain fog I didn’t even know it was there. I told you I would devour this web site you said the opposite….You were so right, but I cant get enough Thanks.
@Mark regarding IF (#354)
I eat two meals on weekends, but on weekdays it is too hard, because it is 12 hrs between breakfast and dinner plus thinking about food gets in the way of thinking about work. So I do eat a small lunch in the office and don’t have unnecessary thoughts about food.
I think I have all the signs of being LS, however, I am also a little confused about what to look for in regards to sweating. I have never been one to sweat much, even in the heat of summer. I only seem to sweat if I am outside in extreme cold, but only if I am in a snowsuit and I am shoveling snow or climbing up a sledding hill.
Until I started the CT protocol, I had a difficult time drinking much water. Now I am drinking at least two quarts of ice water a day and am enjoying it immensely. I am not sure what I should be looking for in my case.
I don’t sweat, I don’t use deodorants and I don’t seem to give off any offense odor.
a fine article in Nature on Joe Thornton’s lab where they recreate cell receptor evolution of ancient times ie (pre)cambrian. http://www.nature.com/news/prehistoric-proteins-raising-the-dead-1.10261
@john a great article on the power of epigenetics……for sure.
Dr. Jack ~ CT just rocks, and well, today I cried. Yep, I cried with the sheer joy of feeling the best I have felt in at least 20 years. I thought so this is how one is supposed to feel, strong, good, happy, on point, able to function without body distraction. I also noticed something short of a miracle and called my RA doc of 24 years to tell her but couldn’t get past the gate keepers. I have had serious elbow contractures in both arms (50+degrees) forever that have never budged, well, today my left arm is way straighter and closer to 45 degrees and you have no idea how hopeful this makes me feel after a lifetime of believing RA is a disease that cannot be cured and my only hope was to take drug after drug that ruined my immune system further and further and many of them nearly killed me as well with the side effects. RA doc is history.
I thought the paleo diet rocked until CT was added into the mix. I am hoping over time if my body is left to function as it should with proper nutrition and now CT perhaps it will start to reverse a lot of the damage to my joints. It can happen.
Do you think it can have any helpful effects for lung damage categorized as bronchiectasis that was caused by uncontroled infections? That would be my holy grail. TY again.
@Cecile based upon what I have found and observed I think a lot more is possible when we invoke the Ancient Pathway using the paleo diet and cold thermogenesis.
P.S. the above comment was written in a darkened room wearing my amber blue UV blockers with the windows wide open! I am now sleeping like a champ and rising with the sun like a kid again.
@Cecile Awesome! I am very very happy for you!
How did I know your big announcement would be on April fools Day?
@Jack – I’ve been noticing something that happens from time to time in the evenings, say around 9 – 10 pm. My ears have been getting red and warm and my hands an forehead get warm as well. Actually I get kinda hot to be honest and have to crank up the AC. Is this a normal reaction to CT even though my body temp is still low at about 96.5-97?
Also, how long did it take you before you stopped the gasp reflex when entering cold water. And does that have to disappear in order to be truly cold adapted? I ask because I still gasp pretty good when I hit the water.
@The Kid it is normal…..and my reflex stopped after a few days
Here is an interesting TED talk about Wim Hof:
http://www.youtube.com/watch?v=L9Cgaa8U4eY
Dr. Kruse, my father, as I mentioned before, has amyloid proteins in his kidney & his question was whether you had seen remission of the amyloids with the introduction of CT. He started a keto diet & CT last week with great results as to how he feels. Thank you as always.
Dr.K, I try to start my day with being grateful.
Thank YOU for being and doing! Have a nice day today!
Dr. Kruse,
By differentiate,I meant can you tell who is posting if two users have the same name? I assume you can byt he email address. I just want to be sure but I will use my full name going forward.
Dr. K,
I’ve posted before under the same e-mail address.. I have T2DM.
I started CT on Mar 6th and continued up until the last 2 days. I worked into 15 minute showers twice per day @ 52F. Thermometer says skin temp does reach the water temp also. I stopped CT yesterday, because:
– Can’t sleep well.
– A couple of terrible workouts where I “regressed” in strength.
– Pretty bad lethargy during the day. The lethargy has been progressive since about a week after I started CT.
– I’ve gained fat, or at least subcutaneous fat. I’ve been using the same BIA scale to track BF, so this isn’t subjective. Two months ago I was at 12 to 13% bodyfat (BodPod, Skin Fold), and I have even eaten less during CT (I log that too). Already by this morning, my BIA measurements have dropped 2%, comparable to before CT and the lowest since starting CT. Subjectively there’s a noticeable difference in the mirror (I can see the faint outline of my abs again for example).
– Beard growth slowed down after the first week or so of CT and seemed to immediately accelerate the last two days. This is obvious to me b/c I could get by w/o shaving every other day or so during CT.
Prior to starting the meal timing part of the Leptin RX (also started on Mar 6th), I’ve been following a low-carb paleo template and the rest of your recommendations even down to using krill oil and most of the rest of the supplements you recommend, like vitamin K2. As measured by fasting blood glucose, by far the greatest difference has been the advice to eat an early dinner w/ no snacks after dinner. That makes an immediate difference. If I eat a late dinner or snack, my FBG is 115 to 125 the next morn – if I don’t, it’s under 100 (it was actually 82 this morning, lowest yet, the 2nd day after stopping CT). I’ve actually been experimenting with the meal timing and so far at least, have been able to confirm what’s effective for me and what’s not.
I lift heavy twice per week and do HIIT one other day, so nothing brutal there. No other great stressors.
So far by both subjective and objective measures, the Leptin RX is working great, the CT not so much.
Have you run into results like this before? Any advice on making CT more effective for me?
Thanks.
@Dave since your t2d the one thing that could be behind this is a high E2 level…..So I would say if you want to see if I’m right get HS CRP, E2 level Highly sensitive, SHBG, Free and total T, Vit D level done and a DHEA level done…..this will get to the bottom of it. If those are all negative then I would move to get more detailed things looked at like BPA and heavy metal toxins…..my bet is you have either a high E2 level of you have a ton of BPA or heavy metal exposure. Those are the people who react like this…….
Thanks! I will look into further.
Hey Doc! Awesome post and for some reason they are getting easier to follow 🙂
Major question for me: Would Low Dose Naltrexone (LDN) interfere with any of step 20 or 21? I guess it would depend on if it competed for receptors with the leptin?
Apologies if you have answered this in another spot, maybe the leptin rx. couldn’t find anything.
@MGH I dont know the answer but you can try it out and see how it goes…..CT should decrease the need for LDN eventually.
Dr. Kruse,
In order from worse to less worse, would you say my list is right?
The ones I can think of (and if you could think of any others, would appreciate
Trans-Fats
Omega-6 (in excess to Omega-3)
Fructose (HFCS)
Sucrose
Lectins (in grains)
Gluten (in Grains)
MSG
Stimulants (like Ritalin)
Anything else?
@BenG light…..technology at the wrong time is as bad as any other on that list.
That list in reference to what causes Leptin Resistance.
DR K
I’ve been spot CTing for close to 7 weeks. The only major hiccups – were week 2 & 3 when I had some crazy hormones, and my TOTM came a bit early. I’ve slowly shifted to CT Baths 2-3x/week in the am/mid day when I can, as I’ve discovered night baths = crappy sleep.
Now, Suddenly in the past 5days I’m waking up btwn 1-3am, and Can’t get back to sleep and waking up has suddenly become difficult! Going to sleep initially at night is ezpz – I’m asleep w/in minutes of my head touching the pillow.
I know my internal engine is working – as I’m getting all hot most of the time…but suddenly on Monday – I couldn’t get warm I was cold all over- however even a luke-warm shower was too hot.
What’s going on? Why am I suddenly not able to sleep, and why do I suddenly feel cold, but luke warm to cool showers feel HOT? Am I just peeling back another layer?
Any insight would be appreciated!
@Shijin you have a pretty specific condition that I bet is causing a slow steady toxing dump……..PCOS is a real issue and takes a while to work out…….but CT works best for it longer term.
Hello,
CT is going well. I’m sleeping really poorly (<2 hours last night) and hit somewhat of a a plateau in the tub (20 mins 48 deg). I feel like this is part of the process and if I stick with it I shall progress.
Somethings I've noticed:
-Looking much more ripped despite very poor sleep.
-After I get out of the tub I have ringing in my ears for the next hour.
-My patulous eustachian tube is acting up much less
-When I do sleep I often have really vivid dreams ending in orgasm. Not much increase in day to day sex drive though.
-Eating way less
-Hair looks awesome
All the best to Dr. K and the rest of the soon to be cold adapted mammals.
Andrew
@Andrew keep us informed.
Dr. K, You recommended a Leptin Rx married with Leaky Gut Rx and CT for me as I am osteoporotic with severe food allergies. Also have had Nissen procedure but don’t know if that damaged the Vagus. Have been eating Paleo for about 6 weeks, Keto for 2, want to start CT soon. Since you don’t use coconut oil on CT but do for the Leaky Gut should I discontinue the Coconut oil? Also how low could I go on calories and still be OK. I eat about 100grm protein per day as I can’t get much more than 50 in for breakfast, CO brings up calorie count, can’t eat bacon as all organic bacon I have found has celery used in processing which I am allergic to.
@Vicki yes I do.
@Dr. Kruse: In CT7 or 6 you mention that it’s normal for people to get tired after an orgasm.. which of course I have noticed in everyone except myself. I always thought it was just something weird to me, but now I wonder if it is a sign of something wrong with me. Whenever I have an orgasm, no matter the energy expenditure (sex vs masturbation), I am instantly hungry and often am more awake than before it. I could have just eaten and be completely full, but after orgasm I will always be hungry. I will also usually be instantly more awake.. tho not always. On the Leptin Reset I can eat dinner and not be hungry all night even with an early dinner at like 5pm.. but if I have an orgasm I will be instantly starving and have to go to bed hungry (I don’t want to mess up the reset w/eating). I’ve never known anyone else with this same reaction male or female. Do you have any idea why this would happen?
@ Erica……people who have this issue usually have a sleep problem too. I send this issue to the sleep labs.
Dr. K, referring #394 – #396:
Looking back in medical records over the last year, all of those except for BPA and FT have been tested for, and they were negative except for a moderately high copper result (urine test). In addition I did a SpectraCell nutrient test and that actually looked great.
I did a mild chelation for the excess copper w/ Zeolite and the results were still positive, which that Dr. explained could be due to adrenal problems. The ASI came out OK, _except_ for a somewhat inverted cortisol curve (although my DHEA was top of reference in that).
From what I gather that seems to leave a potential problem w/ BPA, which I wouldn’t doubt in the least considering my prior diet and habits. Certainly can’t hurt to try and cleanse that out of my system..
What is the best method to cleanse BPA exposure?
Thanks again for all of your time!
@Dave the copper issue points to a liver issue……could be a minor one like Gilbert’s syndrome……but it could mean a bad detox pathway in the liver. With high copper’s (rare) I send people for function testing to look for the cause. Get in to see someone to find out why……..if it’s Gilbert’s no issues……..if its not then find out. It could be a SNP too. I have a patient who found out he had a SMP that raise his copper and his family historyw as loaded with people with basal ganglia strokes…….this is why. The carriers of apo E4 allele also have an issue to worry about with copper. Apo E4 is called the ancestral efficiency allele by people like Grace, aka, Dr. BG. These people appear to exhibit higher concentrations of heavy metals with simultaneous decreased liver detoxification of trace heavy metals. BPA is brutal to remove once ingested A blood test can be done to see exposure. I have seen a lot of big BPA numbers in younger paleo men…………no one has a good way to cleanse it from our fat in my view. CT is the only way I know.
Hi Dr K. Sorry to ask kind of dumb question, but are the benefits of CT experienced the time we are doing our CT or the time after or both? If being cold afterwards is h-e-double hockey sticks, are we losing the benefits by doing something to warm up? I mean, is the point of CT to be cold or for the body to figure out how to warm itself up? Thanks
Dr. Kruse..
On the Detox issue from CT.
If someone has a lot of fat to lose. Let’s say 50lbs.
It’s probably unlikely, that in one month they will lose more than 10lbs. I know you mentioned that it takes 4-6 weeks of detoxing before things start clearing up. But assuming they still need to lose the remaining 40lbs of fat, isn’t that remaining 40lbs going to also have a lot of toxins and cause side effects until the person get’s lean? Or am I missing something?
@BenG no……the reason is the liver’s detox pathways get upregulated…..unless you are trashed. FM and Lyme patients are those patients.
@Jack – I want to help a friend. He is 48 and has been told he needs his gall bladder out in 1 month. His liver is enlarged and inflamed. He is also type 2 diabetic, diagnosed about 2-6 months ago. Is there anyway for him to avoid this surgery? Will Paleo + aggressive CT make it so that in a month his docs will at least pull back from the surgical option? Is there anythin specific that can be done now to intervene?
Thanks so much.
http://newsinfo.nd.edu/news/29908-alumna-haley-scott-demaria-to-deliver-2012-notre-dame-commencement-address/
In addition to Ice baths and spot CT, would it make sense to consider Zeltiq procedure to speed things along? Is the procedure unnecessary for one who is doing cold baths and spot CT, or do we not talk more about it as an option simply because of the cost? I am trying to understand if Zeltiq has been mentioned only as an anology or if one should consider it assuming we have the $$. Thanks.
I have been on leptin reset for 3 months(strict keto actually,no eggs, tons of CO and Ghee and grass fed meats and….you get the idea) and CT for 7 weeks on saturday. Using blue blocker glasses if I have any light after 7, asleep by 9 up by 5. Sleep well
Having some issues, just wondering if anyone had some input for me…
I am late on my TOTM, by 2 weeks. I am never ever late unless I am pregnant, I know the minute I am, and I know I am not. I mean I know 28 days down to a few hours when it will arrive.
As well in the past week I am getting(dark) facial hair, I mean what? I am so strict about LRx and I know my hormones should go wacky, they did week 2/3 of CT, pmssy and acne and greasy and all. But this is awful.
I am in Canada, and my Dr will not run tests, just had them and they were ‘fine’ and I just cant afford a naturopath and hope that they may run them. I am freaking out, more so about the hair!
Sorry this isnt really a positive post, but I am thinking this is a deal breaker for me I just dont know what to do…
Thanks
@Honey Bear you definitely need testing with that history. I dont think CT has anything to do with this at all.
Should body temp drop as low as 91 deg f after 10 minutes of immersion in 50 deg water? Thanks, can’t wait for the 31st.
@Bob S 91 core temp is not good and usually means death……but I doubt your core is that low cause youre posting here.
Yup, it’s definitely not a linear process, but am in the ‘linear’ good part of the process again.
I am currently having to face some criticism on what I eat (I don’t put it in anyone’s face, but they see what I eat on a daily basis), which is then followed by snarky underhanded comments—well my internal changes from doing this + CT are of course not obvious to the public, but I FEEL so much better already, and I am quite confident the more physical appearances will soon follow.
I want to say to them, “Come talk to me when I’m 60, 70, 80, 90 ++++, THEN criticize what I eat”. That is sometimes the hard part, but I can’t bother listen to their foolishness when I KNOW I’m doing the right thing. Btw, said person is not feeling well right now and hasn’t been feeling well for a while…they’re just not used to their new more physical demanding job… Well guess what I had to walk 16km yesterday w/ NO soreness or tiredness RIGHT before my TOM…followed by MINISCULE cramping this morning which one advil took care of and is completely gone already! This is just after a few weeks of CT, and only a WEEK of actual being serious w/ keto seafood as in…no ‘treats’ or whatever hell non sense in between.
I am not making light of this at all.
Dr. K, you know it and I am so grateful for your kindness and compassion to all, your sharing of knowledge and just the love for life. Thanks.
I dropped from the high desert to LA day before yesterday and the clouds blew in just now. I had to lay down for a nap a couple hours ago and now I am burning up from the neck up. What gives?
Never mind. I remember I had some sweet onions and hyperventilated a few times this morning.
Dr. Kruse,
I’ve been following the CT plan now for almost 3 weeks with ice cold soaks in the tube 5 nights per week. I live in WI so the water is very cold this time of year. I usually only last 15 minutes before I start to shiver hard. My diet is very clean in terms of paleo.My sleep is perfect most nights. Here is my question… I’ve been checking my BF every couple of months for the last year. I generally am 11-12%. I checked my BF today for the first time since starting CT and my BF has gone up to 15%. What gives? Any ideas why I’ve increased?
@Mike it is not from the CT……if your EFA ratio is 4 to 1 or above. If your EFA levels are below 4:1 where deep sea mammals are fat biology does change……so if you are using a lot of fish oil stop. Add Krill oil instead or add some omega six from olive oil and nuts. My advice is to test your 06/3 ratio’s to really get the cell membrane ratio’s perfect. 11-12% fat levels require good EFA’s ratio’s. Most people using CT have higher 06/3 ratios and CT shreds fat because of its affects on leptin and fat cells as I laid out in CT 6 and as the plastic surgeons from Harvard University have expertly laid out for Zeltiq.
Dr. K., my daughter just called saying she is in massive pain b/c her Dr (DO) told her she jarred her coccyx during walking over uneven ground in such a way that it compressed I guess into the vertebra. She has a 13 mm herniation between her L4 and L5 with no fluid. plus one other she couldn’t remember. Just above the coccyx she has a congenital extra vertebra which meant she couldn’t accept the invite to Westpoint years ago because she couldn’t do sit ups. So far she’s decided not to do surgery but her movement is becoming more and more limited as she gets older. She’s applied for disabilty and hasn’t worked in several years as an EE. Is this something that the Leptin RX and CT can at least moderate? I suspect she has leaky gut as well. She had the genetic test for gluten intolerance a couple of years ago. I told her about your website but she can’t use a computer much as it compromises her neck and back. I could bullet point the diet and CT for her to print out or read briefly from the computer if you thought that this was worth a try. She lives in Denver and has access to the best medical care. Thanks for your thoughts and I’ll be watching on the 31st.
@Terry F the Leptin Rx and CT are awesome for spinal issues as you will soon learn via my TED talk
@Dr. Kruse: Could you tell me how that relates to sleep? What kind of sleep problem? I don’t understand.
@Erica oxytocin is released in an ultradian rhythm as I mentioned in this blog. But humans also release post orgasm, and women after nipple stimulation. The release of oxytocin is coupled to the adenosine system I mentioned in the CT series that induces sleep. If one has an altered sleep signaling system……ie, a defect in oxytocin release, adenosine signaling, or in the proper development of sleep cycles (narcolepsy) you can get your problem. My bet is that chronic cold exposure in dark light will eventually repair this issue in you. I think it is a manifestation of circadian mismatches in you. My bet is those mismatches in you have specifically affected your oxytocin and adenosine signaling to cause the problem.
Dr Kruse, we know that CT can cause detox; can that detox be the cause of raised hs-CRP levels? If so, how long should one wait before retesting hs-CRP, given that CT is an ongoing thing?
@Coriander it can raise HS CRP…….but it is short lived as long as you apply the CT consistently once you start. If you go too slow or stop too often it will prolong the detox.
Thank you Dr. for your response regarding increasing BF from yesterday. I’ll pick up some Krill oil today! My wife also suggested that she thinks the BF increase is from consuming too much coconut oil as I add it to my coffee and cook with it.
@Mike Tell the wife I doubt it…..CO shreds fat.
Core temp must not be that low, but the oral thermometer says 90.9 post ct down from 95.5 pre ct. Something to be concerned about?
@BobS nope. I think I get a new thermometer.
Dr. Kruse,
The Mrs. has been complaining about constant itching on her bottom for a long time now. She said it has off/on since she was a kid, but recently, it has been more frequent and intense. The PCP originally gave her cream…didn’t work. Referred her to a colorectal general surgeon who looked at her today. He said he didn’t see anything other than a small ulcer, but nothing to indicate the problem. He said coffee and dairy can make it worse. He gave her a different cream and told her to come back in two weeks and if no change, he’d biopsy the ulcer.
Any advice on the health/diet side of the equation you could offer that the medical side can’t help with?
Thanks
@BenG this is an easy one…..stop all dairy and gluten exposure and soak her bottom in cold water……my bet is within a month her bum is better.
Thanks Jack, will try a new therm, but first i’m headed for the pond.Have a great day out there in Nashville.
@Jack: Thank you for explaining. 🙂
I don’t have narcolepsy thankfully. I will say this has happened ever since I remember so since becoming sexually active at least. I don’t know if this is a mess up in sleep cycles.. but I usually have a lot of dreams I remember. I also can wake up at night and as soon as I roll over and fall asleep I instantly start dreaming. The better sleep I have with no wakings in the middle of the night the less I remember of dreams if I remember them at all. Could sleep apnea cause this problem as well? I don’t think I have SA, but I’ve never been tested. Well hopefully, the cold fixes this eventually. Because being hungry after orgasm is annoying at this point.
Dr Kruse I understand that cluster headaches tend to have a circadian presentation due to its relationship with the hypothalamus. Could the rewiring of the hypothalamus from the leptin Rx help with this condition?
@Chase I dont see cluster headaches in practice but neurologist do. I think the Leptin Rx and CT would definitely help
Dr. K referring to #423,
Thanks again, what a wealth of information you have given me! There is a good functional medical Dr. I know of in my area, so I’ll take your info., do some research, and prepare for a visit. From what I understand of Apo E4 and given family history, that may be something for me to look into also. The liver detox angle as well.
I decided last night to just continue CT and work through the poor sleep which in turn is probably behind the rest of the problems I listed. It’s not like my ancestors had access to virtually unlimited heat like I do now, and I figure CT is bound to eventually be hermetic and really can’t hurt as long as I’m able to get better sleep. Besides, I like the challenge.. The good news in all this is that CT is definitely having some sort of effect on me biologically so there’s something going on.
Odd but given the negative sleep reaction I have attributed to it, I still do actually enjoy the CT other than the first 5 seconds or so stepping under the shower head 🙂 If temporary increases in body fat continue to be a problem after getting my sleep figured out, I’ll just have to work through it with a little more HIIT and/or some IF.
Thanks again.
@Dave I think this is a good plan.
@Dave: You mentioned possible adrenal problems, with an inverted curve. Has anyone addressed this issue, as I would think it could cause sleep issues and contribute to some of your other symptoms. Maybe not the sole problem, but a big one in need of attention?
Jack, can you clarify the comments regarding EFA ratio of 4:1 above? Are you saying that 3:1 or 2:1 would make fat loss with CT difficult, and more o-6 might actually help? Also, how does using krill oil in place of fish oil change the 6:3 ratio in a beneficial way? It would seem both fish oil and krill oil ultimately are from the same source and both would increase the o-3 side of the equation. What am I missing here? Thanks!
@Rodney I think they are clear…..below a 4:1 ratio maybe a problem. Many in the paleo world are below 4:1 and this favors bleeding, hormone changes and it changes how fat biology works in mammals. It fosters more subcutaneous fat and not less. This is why you want to be tested for the ratio and make sure your EFA ratios are ideal for humans. 4:1 to 6:1 is the range I like. And yes I do use olive oil and nuts to get people with low ratio’s back up. If you read my blog about fish oil written around AHS 2011 at UCLA I say it clearly there. It appears not too many people read it. We have to have proper 06 signaling for life. And 4:1 is best for humans……….When 06 levels are too low other sub optimal things happen to us…….water based mammals are adapted best to lower 06/3 levels and not land based mammals……..it has to due with cell membrane signaling.
Dr.Kruse, I can’t wait to view your TEDX speech. 2012 really feels like a transformational year on many levels.
Reading your blog has helped me appreciate my passion — organic (though not certified) farming. I have a CSA (Community Supported Agriculture) of about 120 members and growing. I am always advocating eating seasonally and the importance of thinking about nutrition on a year-long basis rather than day to day. Maybe just a few bits of something is all we need for the year. For example, we include many “weeds” as part of the vegetable share — callaloo (wild spinach), wild chamomile, dandelion, ramps, purslane (one of the few plant sources of omega 3s) and spring-time grass fed butter where cows have been grazing on the first spring greens. I always tell members if we were paying for nutrient dense foods, kale would be the most expensive food.
We also make compost tea from our own compost material and spread it on all the fields 3 or 4 times a year to stimulate micro organisms (it’s always the little things that make the biggest difference!) And we don’t coddle the plants (with fertilizers or greenhouses) because we think that plants that have to work harder to survive and thrive are more nutrient dense — and taste better.
Also, I never wear sunscreen! (Although I do wear hats and sometimes long sleeves during the heat of the day). I find I only get so tan and then no more. And to work during the hottest part of the day, I have a long sleeve cotton shirt and I soak in cold well water and wear it — it’s like a personal ac.
Plus, I very, very seldom watch TV — have no cable and just watch an occasional movie. My two sons do have video games but whenever they got where they were playing too much I would simply put it away for a few weeks. This helped them to self-monitor their usage. We also home-schooled our kids — but that’s a whole nother post!
So, thanks again, Dr. Kruse. I love you cookbook and am working on CT.
@Jake thanks for the kind words. As I said in my Jimmy Moore Encore podcast in January 2012 was going to be the year I unleashed Factor X and in the first 3 months…….the ride has been interesting. But I am really just starting things up. This Saturday I am raising the stakes for poker.
But Jack, I understand it will be WEEKS before we have an opportunity to see the TedxNashville video! Please tell me I’m wrong.
@Suni I have no control of that…….I am going to give my best 15 of my life…….that I can say without reservation
Jack, thanks for referring me back to the fish oil blog. The most recent comments there really help clarify things even more. My 6:3 test is due back in a week or so, but I had not remembered you talking about lower ratios (2:1 or so) leading to increased sub q fat. Hopefully now I will know how to interpret and act on my test results when they arrive.
Dr Kruse,
Is Orthostatic hypotension a sign of too much w3? Been getting dizzy standing up I also noticed when I had my blood taken last week there was a lot of blood under the skin–bruise looking. It’s still there. Also bleeding longer when I tested FBG from the glucometer I’ve been taking two fish oil capsules daily since January, and not much w6 since the LRx. Even before the LRx, I didn’t eat much w6 since I knew it was bad from the good old days of Udo Erasmus’ book “Fats that Heal, Fats that Kill” , so I only use to eat olive and flax oil and avoided industrialized seed oils, so I was probably starting from a lower w6 ratio than most people. Should I up the nuts for w6? (without testing)
@BenG you should know the answer……test always.
@Rodney regarding #443,
Thanks for your response. I think the inverted cortisol curve is / has been a problem and I was hoping that CT would help to solve that by improving sub-optimal sleep.
So far the opposite has occurred as mentioned, at least w/ sleep. The ASI is relatively inexpensive so I will look at doing that again also.
Both the Dr. and the ASI test interpretation from the lab basically said the inverted cortisol curve was mild but something to watch, and nothing else indicated adrenal exhaustion yet.
I’m glad you and Dr. K. brought the 06:03 ratio up again.. I should finally get that checked as well. I may actually have too low of ratio which might be part of the subcutaneous fat increase (but still I think poor sleep is the majority of the problem).. I’ve been taking quite a bit of 03, for quite a long time, along with eating pretty clean, and only recently cut O3 down to be more in line with Dr. K’s KO recommendations. Ummmm, I’ve been craving almonds and cashews lately too, and almost can’t stand the thought of eating fish although generally I love it.
@Dave poor sleep is a huge problem……no doubt.
Hi Jack, could the first six weeks of CT cause an increase in liver enzymes AST ALT and an increase in TSH?
@Bob S yes it can because your liver is working over time to clear toxins…..and TSH will be affected by a more oxidized plasma.
Well, me thinks I bragged too much. Right after the post I was unloading a new piece of farm equipment and smashed mt left index finger. I broke the tip of my finger and had to get 7 stitches, a tetanus shot and 7 days antibiotics (and requested vicadin). I think universe is telling me to slow down and get into a cold bath. Dr.K how much should I aim for (cold bath)to assist in speedy healing? Thanks…
Jake put ice packs right above the injury on the wrist and forearm and watch what happens…….more you use the better you do……add 500 mgs of Krill Oil to the mix too three times a day.
will do. thanks. i’ll keep you posted.
Dr. Kruse, do you have your surgical patients ice their incision wounds? Thank you.
@Larry everyone does it if they are my patient………
Dr. K
We need a “Sleep naked on top of covers” protocol. Sigh.
@cooling web I hope not…….
@Jake,
I have to say that’s awesome you are farming like that. I wish I wouldn’t have cast myself this pair of golden handcuffs earlier in life, so I could do the same. My wife and I also home-school and are able to raise chickens on the patch of land we do have 🙂
Good luck w/ the injury.
Dr. K, question and a testimonial for you.
Question: I’ve started having really vivid, awesome dreams since doing the leptin RX in the morning minutes before my alarm goes off. It’s like going to a movie theater every night-I love it. Some websites say this is related to having low blood sugar and talk about it like it’s a bad thing. I’m curious if we know what causes this, and if it is harmful?
Testimonial: I’ve got my energy back! I’ve been battling fatigue for a couple of years, and downing soda after soda and sugary snacks to try to stay awake. Since starting the leptin RX and CT, I have now been able to finally quit soda. The withdrawal was *tough* but so wirth it. Best of all, I have tons of energy and now don’t need any caffeine. I’ve switched to decaf green tea and water to drink.
@Jack That is what I figured but I just like to verify. You truly are a special man with the way you are helping people & I have a feeling that number is going to grow exponentially this year. Keep on rockin’!
I’m wondering (sorry if I missed info) if your next blog will give us a glimpse of what your TED talk is about? It’s all so interesting and I can’t wait to hear more… my life revolves around reading and digesting all this info you’re sharing–literally and figuratively. Thanks!
@paleoNana The TED talk is all about my three thought experiments that became real life experiments to disprove the Ancient Pathway exists in humans……..and I failed all three times. Essentially that means good news is at hand for our species for those who cold adapt.
Jack should I be worried about a couple of my labs:
Ferritin, Serum 398 ng/mL (30-400)
Reverse T3, Serum 47.8 ng/dL (13.5-34.2)
C-Reactive Protein, Cardiac 0.62 mg/L (0.00-3.00)
Vitamin D, 25-Hydroxy 56.5 ng/mL (30.0-100.0)
Iron, serum 102 ug/dL (40 – 155)
Iron Bind.cap.(T1Bc) 353 ug/dL (250 – 450)
I know my Vitamin D3 still has a ways to come up… (I’m eating fairly well, could use more fish, and doing CT nice and slowly.)
Dr. Kruse,
When I initially went LC three years ago, it was to lower my triglycerides. I got them down to 32 for a while. How low is too low with this lab number? Thanks
Susan M.
@Susan low TG below 60 is cooking with gas. It tells us you are burning fat and the liver is in good shape.
Thanks for the response, because I was just a little bit worried about that. 🙂
What changes have you seen in your skin with cold adaption and the dramatic weight loss? I am seeing some good results, but have a ways to go. I’ve had some unusual side-effects of very low blood glucose and hand/foot cramps and numbness (but that maybe more from stopping thyroid meds.)
How do you keep your torso numb all the time (sleep, surgery, etc)?
@DrMommyN you will hear about this soon enough.
“I am kind of boring. Cold dark and sleepy.” Well, aren’t you forgetting sex? Reproduction is rather important, yes?
@Dr MommyN What do you think I do when it cold dark and before I get sleepy? Somethings are better left unsaid ……….but my wofe can speak to that part of the Ancient Pathway herself.
http://fitness.mercola.com/sites/fitness/archive/2012/03/30/hand-cooling-device-for-effective-workout.aspx?e_cid=20120330_FNL_art_1
The copy cat machine is picking it up now…..
I’ve been doing low-carb for 10 yrs, very low carb intake 20-30carbs per day. Take 4,000 fish oil a day plus eat fish. I was disappointed when I read that you thought all post-menopausal women need biodentical hormone therapy. In the scheme of things, why would every women need an outside drug to make them whole? I am 57yrs old and do not have memory issues. I am having frequent hot flashes and trying to determine if this is normal or do I have something wrong? And why in America do women have so many more hot flashes? Not sure if you have the answers and I know this is not your speciality but would like to hear your thoughts…..Thank you, appreciate your dedication to the public.
@Salle With 4K of fish oil a day you must get your 06/3 ratio checked. You might not need the high a dose. Why BHRT? This is just replacing the hormones you lost with menopause using bioidenticals and not synthetics so this is not an “outside drug” it is one every woman makes and needs. The reason why so many women get hot flashes is because for most of their reproductive life they are estrogen dominant. When they go through menopause they lose the balance between estrogen and progesterone and it is even more dramatic because of the E2/prog changes begun in perimenopause. The most common reason for hot flashes are the imbalance between the two hormones, lowered DHEA, lowered pregnenolone as well. I dont think any woman should have to settle for these symptoms.
I see Mercola’s site doing more on this lately. I will post my amazing results from your LS + CT after 2wks after I read CT 7!
http://fitness.mercola.com/sites/fitness/archive/2012/03/30/hand-cooling-device-for-effective-workout.aspx#
As we become cold adapted is shivering eventually replaced by non shivering thermogenesis?
@BobS that is the goal……you will even notice goosebumps when you eat a fat laden meal. When you enter a cold place like your car you will cold adapt as soon as your skin hits the cold. You become a very efficient fat burner.
Dr. Kruse,In trying to implement a keto diet with more seafood and fish(both which I loathe) I’m having a very very hard time dealing with the overpowering metallic taste I experience when eating any kind of fish and or seafood. Is this metallic taste a sign of some kind of physical ailment or a nutritional deficiency? I’ve asked several people and they don’t experience the metallic taste when consuming fish/seafood, so it must be me. I may have to resort to just using Krill and fish oils which I don’t have to taste. If anyone knows what this is, I’d love to know what to do about it.
@L@2be it is eaither poor quality fish……with lots of toxins or it is a Zn deficiency in you. Take 50 mgs of Zn a day for about 10 days and see if it goes away. If not then look at your fish….
I also wanted to ask about good supplements to detox ED with. I’m currently using D-Glucarate, along with Indole-3-Carbinol twice a day. B12, B6 and NAC I can do as well. Would there be any benefit to adding Trimethylglycine to this? Would having additional methyl donors increase the detoxing benefits of the other supplements?
Calcium D glucarate is great stuff for detox with CT. So is I3C along with DIM, B vitamins and mushrooms…..
Ah, Zinc! Yes I am zinc deficient!! I never connected that to the metallic fish however. Thanks so much for that! I’m eating the same fresh fish as everyone else and I’m the only one getting the metallic taste.
@Jack – I have done about 4 weeks of CT. It’s been slow going, I still gasp when I enter the shower. I also have a rash that won’t go away. Quick question:
– Which is preferable: A)a shower that is always 50-55 degrees or B) a bath that is always closer to 60 degrees.
Note: The cold bath I have access to has water coming out of the tap that’s about 55-57 degrees, but the water in the tub is always closer to 60. And I am assuming that my own body heat with increase that temp once I’m in it for a while. The circulation system is an old one, just a tap and slow drain. No water jets.
@The Kid I am very partial to baths over showers……more coverage is better. The rash is a sign of toxin dumps.
Dr Kruse,
“f I fall off the wagon on the LR Protocol do I need to restart the clock? Yes…If you eat Domino’s and a 12 packm your cooked”
How many carbs will throw you off (is the quantity that significant if you follow the other rules (BAB/No eating after 7/No Snacking)….etc?
i.e. is eating 200g-300g of Paleo starches going to kill it after 8 weeks?
Also, If showing all the LS signs……Post-Reset…how many carbs can you eat before damage is done (assuming no glucose, Omega-6, Gluten, Lectins), etc?
Part of the context I’m trying to understand is Skinny LR folk can eat paleo carbs during reset. If Overweight do it, are they cooked if they eat clean like skinny folk, as long or do they simply lose weight slower?”
@BenG depends upon your HS crp and DHEA level…..if youre way out not much throws you off……..because the cytokines increase. If your already tight you can fudge it more. I dont fudge at all.
sorry, meant no fructose, not glucose..
@Dr. Kruse: I’m no where near menopause, but I had a similar question to Salle’s.
I’m 28 right now. So, say I fix my leptin, do ct, basically become pretty optimal. Do you still think biodentical hormones are necessary when I do hit menopause?
Because I would think that if we were healthy then menopause wouldn’t be such a big deal and the change in hormones are obviously natural. Why would we have evolved to have this happen if it was a bad thing for us?
@Erica dont assume that…..most women in the US are entering peri menopause now at your age…..if you get your levels tested you might be surprised by how much erosion of your hormones you have. This affects the oocytes you mature too and why modern women have higher risk pregnancies and higher rates of issues like autism……..its all tied together.
My girlfriend has some interesting problems and I was wondering how they might relate to your CT/paleo theory. Her body temp hovers around 95-97*F and has her entire life. Even when she’s ovulating it doesn’t get close to 98.6*. However she is constantly cold to the point of discomfort even in rooms around 65* without extra layers. Shes an endurance athlete and thought when she started the increase in muscle activity would increase her metabolic rate and fat uncoupling (speigelman’s work on irisin etc.) but while she sweats much more there has been no change in core temp or ‘feeling cold’. She tried paleo for a couple months and was constantly fatigued and had difficulty sleeping/recovering after any workouts even very light ones. She isn’t on any hormone supplementation or birthcontrol (we switched to natural planning a few years ago so she’s been off anything for at least 4 years) and has a very high sex drive except when she was eating paleo and constantly tired. She tried CT briefly but didn’t seen any benefits and just the thought of cold water stressed her out… although I think she was just being wimpy… Is CT/paleo just not right for her? We are trying to do this together but it’s tough for me to encourage something which doesn’t seem to be helping. Is it possibly she has some deficiency (protein or otherwise) causing cold intolerance?
@Matt her endurance exercise is burning her adrenals out……..she will never do well until she stops it…….CT and paleo is ideal for her but since she is running her adrenals on overdrive she thinks she feels best that way. A diurnal cortisol and DHEA level would probably make her shutter with the implications.
Cherry angiomas appear to be cropping up all over my thighs and stomach. I’ve been discounting it but there are many and some are so tiny – I really think there are a lot more than a month ago when I began CT.
These are signs of estrogen leaving the fat?
@Clabber yes they are. I had tons and they go away eventually when your E2 levels fall.
Forgive me for the follow up but she wasn’t training during during the paleo/CT time… specifically because of what we’ve been reading. So does even moderate exercise (easily less than 20 miles a week running) have the same affect? I only included the endurance training history because I figured she would already be adapted to increased muscle beta-oxidation and triglycerides, not because she was training.
@Matt its about her diurnal cortisol rhythms……did she have them tested? How about just a simple DHEA level to give you a hint? Cortisol/DHEA is a real easy way to tell what is up with her.
Thanks for your quick reply. A few months ago I went to my GYN and she gave me a prescription for Biodentical Hormones. A combination of Estradiol and progesterone; I went home and looked up the brand name on the internet and the first warning sign for the prescription drug was for an increased risk of endomietrial cancer. I feel more comfortable not putting something like that in my body. Just can’t figure out how I can get rid of them naturally. Thank you.
@Salle – check out The Natural Superwoman by Uzzi Reiss for an easy read on bioidenticals. He talks about doses, different methods of delivery, side effects and reasons for taking them. He explains why true bioidenticals are not the compounds that have been linked to various cancers and in fact how they help protect you.
Jack, I just got my omega-6:3 results and need a little advice.
6:3 ratio in blood = 1.9:1
o-3 index = 13.3% (DHA and EPA as % total fatty acids in blood)
I eat salmon 3 times weekly, no veggie oils or nuts, no o-3 supplements, sardines once a week or so.
What do I eat to normalize to 4:1? I was thinking to add nuts, mainly almonds or a smaller amount of walnuts. I also rarely eat pork or poultry due to higher o-6 content, so I could add those back instead of beef and fish all of the time.
How much of the o-6 foods would it take to get back to 4:1?
Thanks…AGAIN!! I have 15-20 pounds of mostly belly fat to lose that hasn’t budged with cold baths so far. Not a lot, but it needs to come off!
Jack, during full immersion, I get this burning sensation inside my stomach. Is that a sign of being Cold adapted?
@miguel no it is not
I had The best CT session yet this morning! Granted I’m still just doing the showers but our water is at 47 so it does the trick. I stayed in for at least 20 solid minutes no flinching or ill effects. It didn’t end there I came up with an Idea to open up the window (second floor as not to scar the neighbors) and not dry off while shaving and brushing my teeth. It was as if I was still in the shower at 34 degree wind hitting me. That is the first time I actually shivered since starting CT. The results of CT and leptin rx are showing.
@Mike…….Go Mike!
Dear Dr. Kruse,
I’m curious about what you do when you are in the cold water for such a long time. Do you have a writing set- up, like a floating desk ??? Listen to music or science podcasts?
Also do you think there exists yellow contact lenses that would block the blue light? Maybe someone would know that reads this blog. I went into a vision store and inquired and the doctor hadn’t heard of anything like it. I would love to wear lenses instead of the clunky glasses 🙂
I’m anxiously awaiting your next big revelation. Your selflessness astounds me and your generosity in acknowledging each and everyone’s large and small health concerns is a gift and blessing for all who have arrived here. I can’t even remember how I came upon your site at the very beginning. I have been following like an addict. I don’t know what I’ll do when I’m cut off if you discontinue this blog. I’m addicted to the new secrets that unfold each day. It’s like a huge puzzle and each day a new piece falls into place quenching my thirst for more knowledge. I think it was a miracle I found you!! I thank you deeply.
Hi Jack, couple of quick questions if I may:
Firstly, is CT still advisable if you have no weight to lose? I’m 5’9″, 140lbs, be good to put on some weight!
And secondly, is it advisable to do CT with a cold or chest infection, or best to hang fire till it’s gone?
Many thanks
@jonnyH If your not interested in Ultimate health, longevity or performance…….no CT is not necessary
If my DHEAs is high for my age (51), how can it be that my estrogen is low? My testosterone is at the higher end as well, my progesterone is good due to a regular supplementation. Only my diurnal cortisol spikes up, the rest is normal.
I cannot make any sense of that.
@Mart Cytokines all affect different hormone pathways differently and there many different combinations that arise that can lead to thousands of different affects to our epigenetic switches……the one thing to remember is that Cold destroys all the cytokines…….so it resets all the hormone pathways……..that is the beauty in Mother Nature’s design. It is her reset button
@ Mart, seems like DHEA is not so dependent on estrogen then…better to have higher testosterone. Of course, best to see what Dr. K says.
Dr. K: I’ll always have in the back of my mind about BRHT for 27 years down the road. It’s interesting concept even though evolutionary women didn’t ‘need’ that, but then again, they DIDN’T have all the modern insults that modern women have, even with Leptin Rx, CT etc. discovered at a later age. I think someone doing CT etc. even at the age ofe 26, still has 26 years of ‘undoing’, so I think you def. make a good argument for BHRT. Also, the BHRT seems like it’s natural and not that synthetic crap that they use.
I feel lucky that I can correct damage from a younger age. It’s funny, that only when I’m haelthy and eating right can I even BEGIN to have positive thoughts regarding having kids (when I’m ready) instead of in the past being SO against it—like afraid. I think I’ll be more ready in my 30’s, but hopefully not after 35. That’s probably not ‘ideal’, but it’s the way life is sometimes and at least I am more optimistic for a good outcome.
Gladina,
Dr.K says we can become healthier at any age. Imagine the results at your age!
My estrogen is waking up … I have a zit forming on my chin! It is either from the supplementation or CT or both!
I cannot remember when it was last time I was getting a zit. Who would have thought that one day I’ll be excited about that :-)!
Dr Kruse,
Thanks for you the previous answer.
With regards to sleep. What’s the main difference between someone who cuts the lights out at 8pm (Daylight Saving Time) and then stays in the dark, and goes to bed later at 11pm vs. someone who stays in the bright light till 11pm, the-n turns out the lights and crashes is asleep within 10 minutes. Assume both wake up at 7am. What advantage does the 3 hours of darkness have if the other guy easily falls asleep as soon as he turns out the lights?
Also, any advice for me…I eat dinner usually now between 5:30 and 7pm. I go dark by 7:30, but I find the dark makes me sleepy. I get sleepy as soon as the sun goes down. Then I end up falling asleep by 9-10pm. The problem is, it’s only been 2-3 hours since I last ate dinner sometimes b/c I can’t stay awake when it gets dark. Yet the LRx requires 3-5 hours from the last meal. Do I just have to eat at 3-4pm instead (very inconvenient)?
@ Erica, I agree with you. I am def. in perimenopause at 43 and I thought that changing to paleo and keto (which I’ve been for several years now) was going to help me avoid some of the side effects of menopause and having to take hormones in the future.
@Dr. Kruse: And now with CT added now to paleo/keto, why do I have to have a future of Bioidentical hormones? I am doing all of this to be optimal, to avoid drugs and medications. I don’t get it. Is taking anything messing around with MOther Nature again? And without constant testing, how do we know that things are becoming unbalanced in another direction?
@Dr. Kruse: I’m no where near menopause, but I had a similar question to Salle’s.
I’m 28 right now. So, say I fix my leptin, do ct, basically become pretty optimal. Do you still think biodentical hormones are necessary when I do hit menopause?
Because I would think that if we were healthy then menopause wouldn’t be such a big deal and the change in hormones are obviously natural. Why would we have evolved to have this happen if it was a bad thing for us?
http://www.dailymail.co.uk/health/article-1207237/How-sticking-feet-cold-water-help-lose-weight.html
The hardest part of LR & CT was having to stop my 5am workouts along with my ocassional evening Crossfit. I now log my ice baths as ‘workouts.’ One hour each day – minimum. My diet has been spot on Paleo for a couple years. NOTHING I did over the past couple years would drop more than 5lbs. In 2 weeks I lost 13 lbs following your program(& just 10lbs to my goal weight). That is the tip of the iceberg – my mind is at top speed, my eyebrows/hair is growing back and fuller, skin is taught and flush and I am more ripped than when I was hitting the gym doing 430lbs incline leg presses! I am logging details of this journey on Sparkspeople. Your discoveries have implications and potential beyond our initial use in health and nutrition. I am thankful that you have shared your theories and am excited to see cross disciplinary application.
Much like you, my father, an aerospace engineer always encouraged linking knowledge across fields – medicine, engineering, philosophy, nature, arts, etc., I have printed your CT series and dropped it off with him. I look forward to the discussions from layman and scientists on your amazing discovery!
@Natalie when your Dad see what I did to prove it……in my TED talk he may well drop. The bootleg copy of the TED is making its rounds. I would suggest you read this thread on my forum.http://forum.jackkruse.com/showthread.php?611-Dr-Kruse-TedX-talk-to-be-streamed-between-4-amp-5pm-today-Sat-Mar-31
Good to see that kind of thing in the mainstream.
Something nice: I haven’t washed my face with soap or used moisturizer in about a month now—since starting CT. I ‘occasionally’ use body wash…but hardly at all. I don’t smell and of course my skin is smoother. My face looks the best. I do have a question though. My hands and lips are dry, but I’m thinking with more time they will begin to soften up as well. It’s only my hands and lips while everything else is baby soft and glowing. lol.
My daughter found a baby turtle in the drainage system. Why does this needs sound so familiar?
1. Cold, swimming water.
2. Cave-like places to hide in.
3. Sun or lamp with UVB during daylight hours. Diurnal.
4. Sun or heat lamp in dry spot to bask.
5. Very little food, mostly fish, insects, greens.
@DrMommyN Life follows a fractal design……..it is Mother Nature’s rules. Follow them and you become Optimal. It is that plain and simple.
@Nathalie, That is quite amazing progress for only 2 weeks. Congrats to you. I’ve also been paleo for a couple of years and I have 12 lbs that won’t budge. Lost them last year on hcg and they came back again. I’ve had to go very slow with the CT, started with the face dunks and cold showers and moved to cold baths with my tap water coming out at 55. I ended up with cold urticaria a couple of times from ice packs and bag full of ice cubes. I guess you must be healthier than most people to cold adapt so quickly. Did you have any symptoms, like some of us have had? Fatigue, inability to sleep well (I am sleeping great), aches and pains, rashes, hives, etc.
Can anyone point me to the iPhone Q app that is mentioned in this post?
Thanks
@Claudia When I found Dr. Kruse site I had already been at zero sugar, high seafood tight paleo diet with all his recommended supplements including krill oil, K2, chlorella for detox,etc for several months via readings and research I had done on my own. I did 2 days of face dunking then 2 hr ice baths by the 3rd day. I believe having been on a diet w/the right supplements for months may have helped this quick adaption because I HATED cold. The big breakfast and not working out was a first. I definitely have experienced many of the negative effects, i.e., early and heavy TOM back to back, skin breaking out at 2nd week, and randomly waking in the middle of the night. I am currently experiencing my first sinus cold in years and nasty stuff is coming out. Had red rash the first week from ice packs. However, I have read every comment in the CT blogs (while in my ice baths) and know this is common. These symptoms are a cake compared to the bruises and injuries I experienced in brutal workouts and competitions that left me exhausted, bruised and fat. I took an Ice bath twice with this cold and feel better this evening. Ultimately, the benefits completely outweigh the temporary inconveniences. I am shocked this has worked so well. You can see my food/ice baths logs under Cron23 on Sparkspeople. I weigh/measure all food/macro/micro nutrients my weight measurements and ice bath duration so I can run graphs against different variables. I have food logged for years – you can also see that nothing has worked before. Some days I logged more than 10 miles a day plus heavy lifting etc with less than 1200cal on keto and I still couldn’t drop a pound. Hope that sharing my experience can help in some way
@Nathalie Good stuff…….CT will help you big time. The deeper you sue the more benefits you will get.
Quick question: what is the connection between n3-n6 ratio and being able to cold adapt? You probably mentioned it, but I missed it. The issue came up on this PH thread:
Thanks!
@Eric S CT 8 will address this.
Dr Kruse, a friend of mine has Meniere’s Syndrome. It’s usually under control, but it’s been really acute for 2 months now. Do you think CT could help? Thank’s a lot!
@Abetterme I have one patient with it and we used high dose Vitamin K2 and ice to side of head and at the base of the front and back of the neck. It did not go completely away but it improved by 85% according to patient. The patient never came back so I don’t have longer follow up.
Dr Kruse, My husband and I have recently started your leptin reset protocol. I am also a new/breastfeeding mother. I was wondering what, if any supplements were safe to take during this time? Also, my son still wakes up to nurse once a night between the hours of 2-4 am. I know my cortisol should never be high at night time and I’m missing out on premium sleep hours. Am I doing this reset in vain? I am ready to commit, however, I still must meet my baby’s needs. Should I quit the reset until he is sleeping through the night? Thank you for your help!
@LaurenS No supplements……just eat a good paloe diet and let the baby feed. You will bounce back……..I would suggest to you that you add one hour a day of meditation to you regimen before you go to bed. It wil help you too. I also would go dark after sunset and use that technique to go to bed earlier your self to offset the baby feeding at 2-4 AM
@Ld Vulvodynia is VERY responsive to a low oxalate diet. I’ve been reading the Trying_Low_Oxalates yahoo forum for almost 2 years. People getting benefit from LO include vulvodynia, autism spectrum kids and adults, and people with interstitial cystitis or kidney stones. My fibromyalgia pain is maybe 1/5 of what it used to be, and I no longer have the fibro fog or mood swings associated with it.
@Lynnet Thanks for the forum reference. I used to do low oxalate and quit after a few months but I’ll try it again. Wouldn’t you know – both 100% chocolate and turmeric are very high in oxalates – they’re now out of my diet.
some of the best change making reading on the planet these days
here is an excellent link on how mood and depression are made worse with biological mismatch created by modern society.
http://www.acnp.org/g4/GN401000099/CH097.html
Dr Kruse, Past few days my pinky is turning White y hands are normally red on inside
not sure if its from a nerve or ? it turns white and tingles. might have strained my hand or my neck was bothering me at the time. It does return to normal color. feels sore
What do you think? put cold above? on wrist?
hmmm so we are going to reset the brain to match the body biology? and then evolution may catch up in a uniform way?
I’m in the Zone reading your blogs The mindset has preceded the physical reset
@JoannH fingers are more sensitive to cold…..above the wrist is a good idea.
I am reading several items from you since this am at 3.. I am still not clear as to what is going to be the need for me to read First to get the Plan.. and second to follow through so I get a total TO DO picture.
The read is good but long and I am interested.. I am on diet for now but I am thinking of after.. I wish to stay down..
Would swimming in cool 80 degree water help? Cold scares me.
Sorry to be so presumptuous as to impose questions.. You are generous.. but where to start and what comes next.?
THANKS..
@Jolanda B Start here…….simple. https://jackkruse.com/easy-start-guide/
Here is a great blog post showing more CT affects in Humans from Everest and talks about the hibernation that some bloggers dont seem to know is present in humans.
http://inhumanexperiment.blogspot.co.uk/2010/03/curious-case-of-human-hibernation.html
Dr Kruse, Thank you for all you do!
It worked! I put a cold pack on my arm above wrist and the blanching of my pinky stopped.
Interestingly, my husband was always HOT and I was always COLD. Now since I have embraced the skin sensation of feeling cold, I no longer need to turn heat on, but my h since losing weight feels cold. he lost 30 lbs in 2 months.
Still wondering: are we reseting brain to match body biology ? Eventually, body will evolve to environment when no mismatches?
Hi Jack,
You mentioned that BP is the highest in the morning. I have definitely seen that be the case and have at times scared me that my paleo diet is not having the impact I would like – but when I measure it in the evening – often after a glass of red wine – it’s in check. What time of day do you recommend to get an accurate reading and beyond CT and paelo are there any supplements you totally recommend to lowering BP? Right now I do 10k VitD-3 and 5G Fish oil.
@Brian remember that this post deals with normal and ideal situation. Most people are far from ideal. When I was checking my BP it was in the AM and right before I went to bed. Resveratrol and turmeric PC and PS are good ones for BP control.
OK………if you made it through this massive blog…….and the 550 comments…….Here is the gold nugget for perserverance.
Google cycloset.
Then read the Prolactin section.
Then become a member on my site.
Circadian biology trumps anything in a paleo 1.0 book any day any way. Trust me……The Brain Gut seires coming soon will prove it.
So you like cycloset for T2D with Hashi’s?
@Dali It depends. Your case would be intersting to run past your doc……if your FBG are still out of whack it could be the last step…….for a big time reversal. I am a fan of cycloset and metformin for anti aging as well. It is rarely used but it works miracles in post menopausal women as I mentioned briefly in CT 7 in the Prolactin section of the blog. https://jackkruse.com/cold-thermogenesis-7/
@BenG #518
I’ve been wondering the samething too. It can get dark pretty early in December in Michigan. If I went by light levels, I wouldn’t be able to work; I’d be asleep by 6p. That means I’d have to eat my dinner around 2p. However, I believe that I read somewhere else on this site that the reason you should not eat after dinner is because elevated insulin levels interfere with the leptin signalling in the hypothalamus. I could be wrong, but I took it to mean that I shouldn’t be eating after 7, maybe 8 in the summer, because I need to leave time for my postprandial insulin spike to wane in order for leptin signalling to occur at midnight.
@Jack (or anyone else with an insight)
Regarding sleep duration, it gets dark early in Michigan during the winter . Is it sufficient to sleep 7.5 – 9 hours per night in the winter, even though there’s 12+ hours of darkness? Also, if my sleep pattern gets messed up on an occassional night, and I can’t get to sleep until 1130p, would that mess up the leptin signalling that occurs from midnight to three? Do you have to be asleep for a certain amount of time, or in a certain stage of sleep, for that signalling to occur? Lastly, does waking up before sunrise mess up the circadian rhythms? The sun doesn’t come up until after 8a in December, and waking up at that time would not jive well with my employer.
@Micheal Moose Previous blogs have talked about this issue many times. You dont need to dinner at 2 PM. Eat when you want to eat…….just be mindful of the light and not to eat too late before you go to bed.
Hey Dr. Kruse, so I saw your podcasts with tristin on primal edge health and the genius ideas I heard resonated with me so much in terms of what I’ve experienced in life so far I had to start reading your blog posts. Anyway, I would love to hear your thoughts on a few questions I have about my current lifestyle if you don’t mind taking the time to answer them. I apologize in advance if I’m asking questions I should be able to answer myself with these blogs but I sometimes get lost in the jargon…
To give some quick context about myself that may help you answer my questions I’m a 19 year old male, fairly lean/muscular ( 5’11” and 147lb), and I live in Whiterock, BC, Canada, by the Pacific Ocean. I’m fairly active pretty much everyday and routinely do intense interval and strength training.
1. I currently only primarily eat one meal a day in the evening. I may occassionally have a few berries, spinach leaves, or coffee cups earlier in the day but I get all my main foods around sunset (at 8pm right now), which mostly consist of vegetables, meats/seafoods, and fruits. After reading this blog I am realizing this may be detreimental in some ways because I don’t always wait 3 to 4 hours before going to bed, but my question is are there going to be any large ramifications in eating this way in your opinion provided I DO make sure to give 3 to 4 hours after eating before going to bed? I’ve eaten this way for the past year because for one it just felt more natural, and second it alleviated cravings and hunger throughout the day and seemed to improve many areas of my life. I also sometimes enjoy a small bowl of rice, potatoes, or popcorn (organic if that matters) at the end of my meal sometimes. Will eating these types of carbs really affect me negatively long term? (I realize season may contribute to this as I live in Canada)
2. I’m fascinated by all the research on the circadian clock cycle and now try to limit as much blue light before bed as possible. I notice you said in this blog though that it is optimal to have 3 to 4 hours of complete darkness before bed? What should one do to try and maximize this? As surely it becomes very hard to perform any tasks in complete and utter darkness. I’ve started reading by dim candle light an hour before bed and I seem to feel more sleepy when going to bed (although I’ve always fallen asleep fairly quickly), but what other things could I realistically do in the hours before bed that wouldn’t disrupt my clock too much?
3. Clean water is obviously very important and being in Canada there is a fair amount of spring water, but often I drink the tap water in BC which could have harmful chemicals. Obviously drinking the spring water would seem to be prefferable but I often only conveniently have access to tap water. Do you think there will be long-term risks in drinking the tap water?
I know not all of these questions have to do with this post specifically but would greatly appreciate any light you could shed on them. I mainly ask the first one because while this way of living/eating appears to be keeping me in fairly good health I ran into a small potentially very harming problem recently. Now I apologize if this gets too personal but it seems you may have better knowledge to answer this than most. Over the last year I’ve noticed a lower sex drive and I initially thought it was just me maturing and because I was choosing to focus more on my studies and exercise. It’s not like the drive wasn’t there but it certainly occurrred less frequently. But then a few weeks ago I lets just say “got involved” again for the first time in a few months and my semen count was WAY down. (probably less than half of what it used to be). I took this as a sign that I may have severely lowered my testosterone levels but I’m wondering what could have caused this. Was it too much intense training? Was it because my dinner with all my main nutrients sometimes fell too close to bed time? Was it my lack of sexual activity? Or was it because I lost too much weight? ( I used to weight about 163lb but lost that over the last year, though even then I was still quite lean so I think I lost some lean mass). I want to know because while it doesn’t appear or feel to be too severe right now clearly something I did made that change, and I wanted to hear your thoughts on things I may want to change about my current style of living.
Also if blue light so adversely affects us at night would it then be optimal to do all ‘electronic’ work as it were, in the morning?
Thanks for any response you can give! Appreciate your time doc! Keep up the good work!
Best regards,
Conor
Conor you need to read the Ubiquitination series right after CT 7. Ubi 4-7 will really open your eyes and answer your questions in detail. Thanks for the kudos. Please share the info to your friends who don’t know how blue light is destroying our biology. I spoke in BC in March at Boucher and have a lot of members from BC. You’ll see them on the forums. Ubi 1 is not good news for those in BC.
Ok, So I think I’m getting things more but help me cut through the jargon a bit doc.
So if altitude is a real problem as said in ubiquination 1 wouldn’t me living by the sea be a good thing? Or when you say bad news for BC do you mean people living in mountain areas? Or is latitude on the earth what you mean? I’m guessing my level of exercise with the protein I get is still way too high relative to where I live if I’m seeing these testosterone problems? It now seems clear to me that those carbs are almost a good thing and what I really need to focus on is less blue light exposure and nailing down natural cycles such as eating a bit earlier relative to night ( although these blogs Don’t help blue light, haha)
And so then is it adrenal fatigue that I’m experiencing from too much exercise and protein intake? Is it possible then to reverse this effect by doing less of these? Or am I still missing something bigger? But then I still need DHA but seafood still has significant protein too… Or should I stop intermittent fasting and eat protein earlier in the day? Does this mean I should Hardly exercise at all because of my location or just less intensely? Your comments about getting addicted to exercise and then leading to possible unforeseen death have me worried a bit…
I would love to move my location to but that’s just not possible right now, but I’m trying to figure out what’s optimal for where I live.
I appreciate your time and quick responses, I hope I’m not coming across too bothersome,
Best regards,
Conor
Conor……you need to dedicate time to read and assimilate. It is not going to happen over night. Adrenal fatigue is a brain stem disease. Read Brain gut 16 and the PVN. The answers are in the blogs if you apply yourself. This site is for self starters.
I understand all that doc and want to take the time, it just isn’t helpful to spend all day reading from blue light when there are SOO many blogs at this point 😛
Brain Gut 16 seem to point me much in the right direction for my situation, but when you say after 5pm for workouts does this mean working out after or before dinner? Clearly this will change too because in BC light cycles change drastically with season but does working out before or after dinner make any difference?
Conor print them out and blue light goes away. Buy my book off of amazon and blue light goes away. When there is a will there is a way. Just depends how bad someone wants info and how far they will dig. I do a lot for free. Now you have to decide what to do next.
Solar frequencies in the evening are similar to morning frequencies, which would be good for steroid hormone production, but humans at that hour lack the huge cortisol spike that sunrise gives. Can the anterior pituitary be stimulated enough by ghrelin at that time? Do steroid hormones get a slight boost for the second time? Dopamine should be on its way down, and the temperature is dropping. CT would be helpful. Perhaps stress from a good fight, city traffic, or hunting scary prey?
Twilight, the “golden hour”, late evening, sunset is a special time for humans. I have noticed an increase in intuition as the sun gets low. Something nice must be happening between dopamine time and melatonin time. Maybe all those energized aromatic amino acids are doing their thing? What’s going on?